Hydrofluorocarbon electrolytes for energy-dense and low-temperature batteries
TL;DR
Hydrofluorocarbon (HFC) electrolytes with fluorine-based ligands enable high salt dissolution, low viscosity, and high stability for energy-dense, low-temperature lithium-metal batteries, achieving over 700 Wh/kg at room temperature and 400 Wh/kg at -50°C.
Key Takeaways
- •HFC electrolytes use fluorine-based ligands to dissolve over 2 mol/L of salt, offering low viscosity and high oxidation stability.
- •Weak F-Li+ coordination improves Li plating/stripping efficiency (99.7% CE) and exchange current density, especially at low temperatures.
- •These electrolytes enable lithium-metal pouch cells with energy densities >700 Wh/kg at room temperature and ~400 Wh/kg at -50°C.
Tags
Abstract
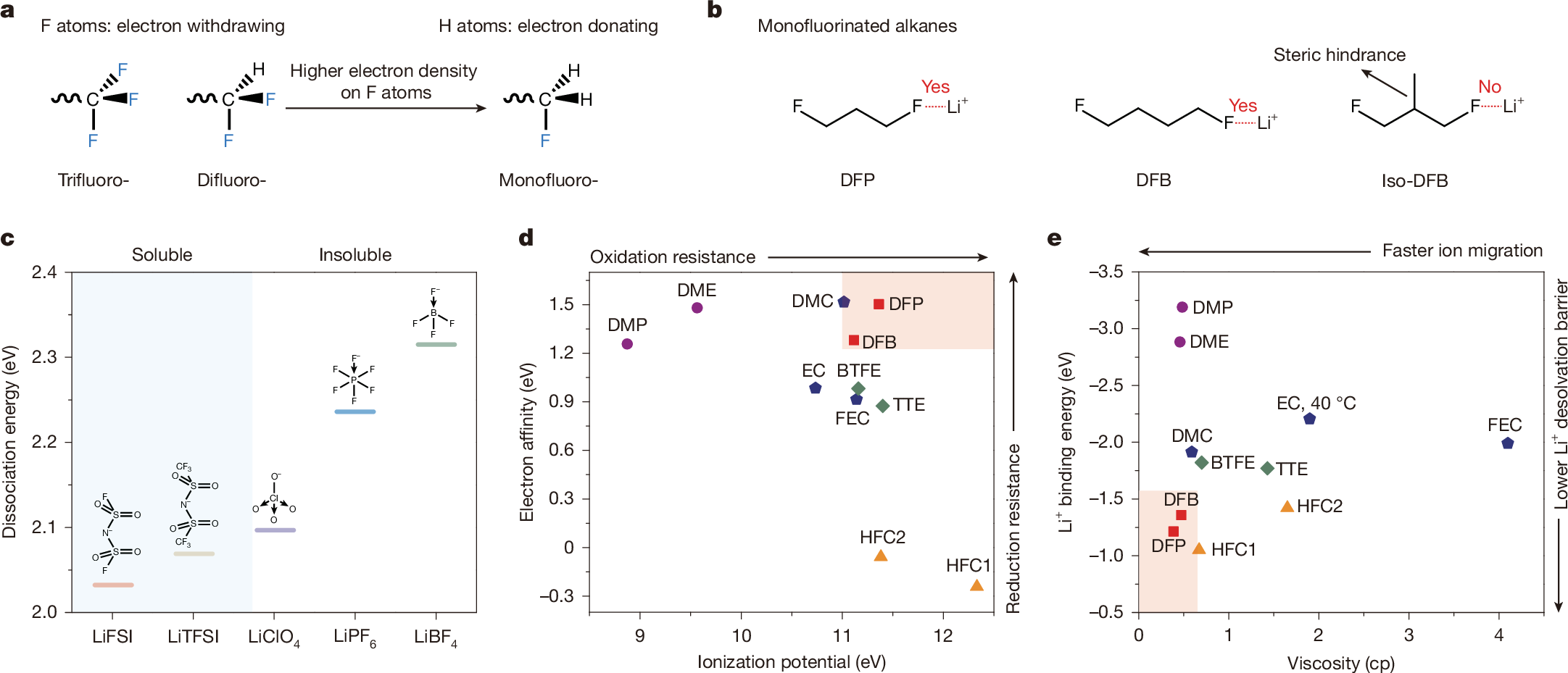
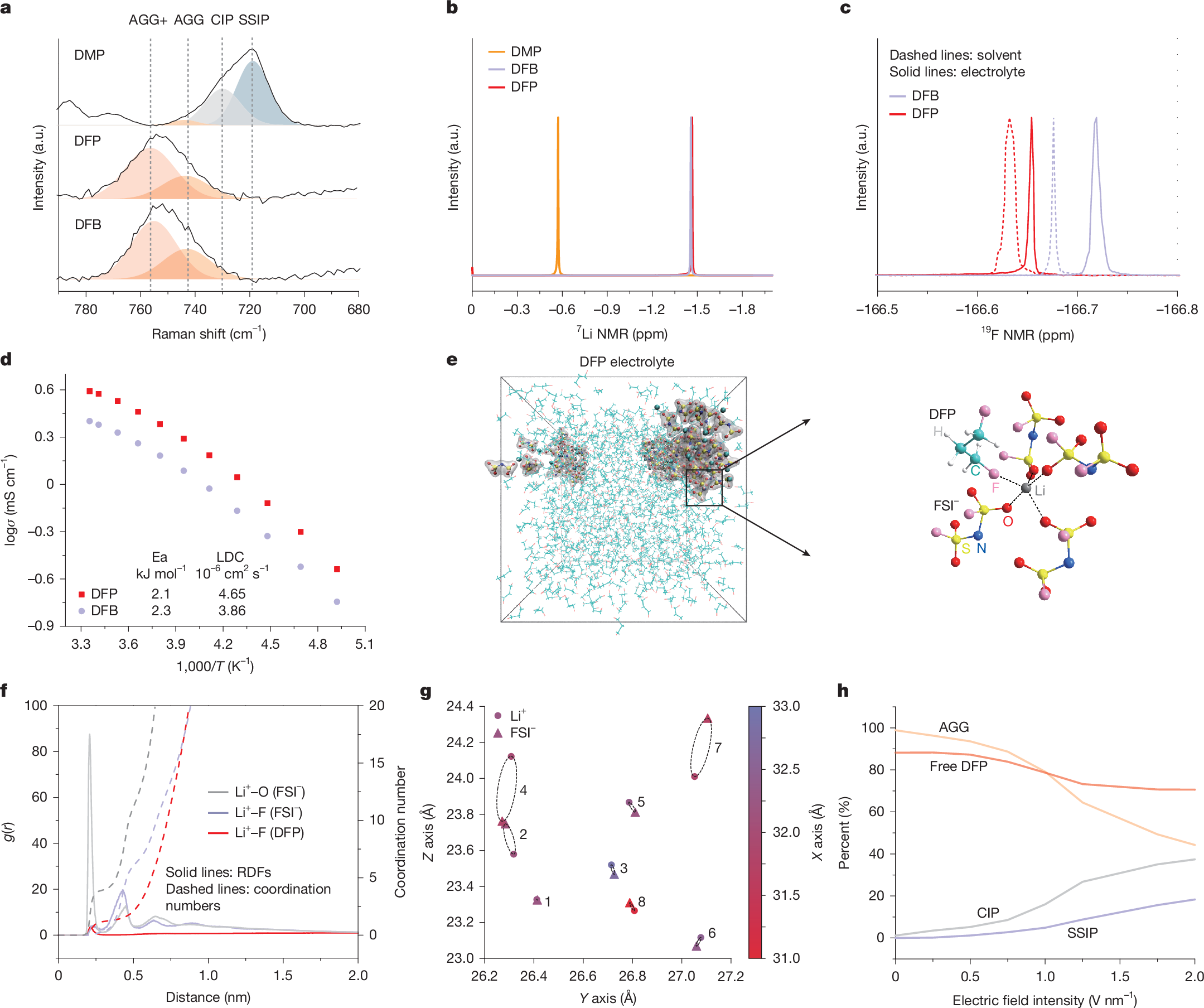
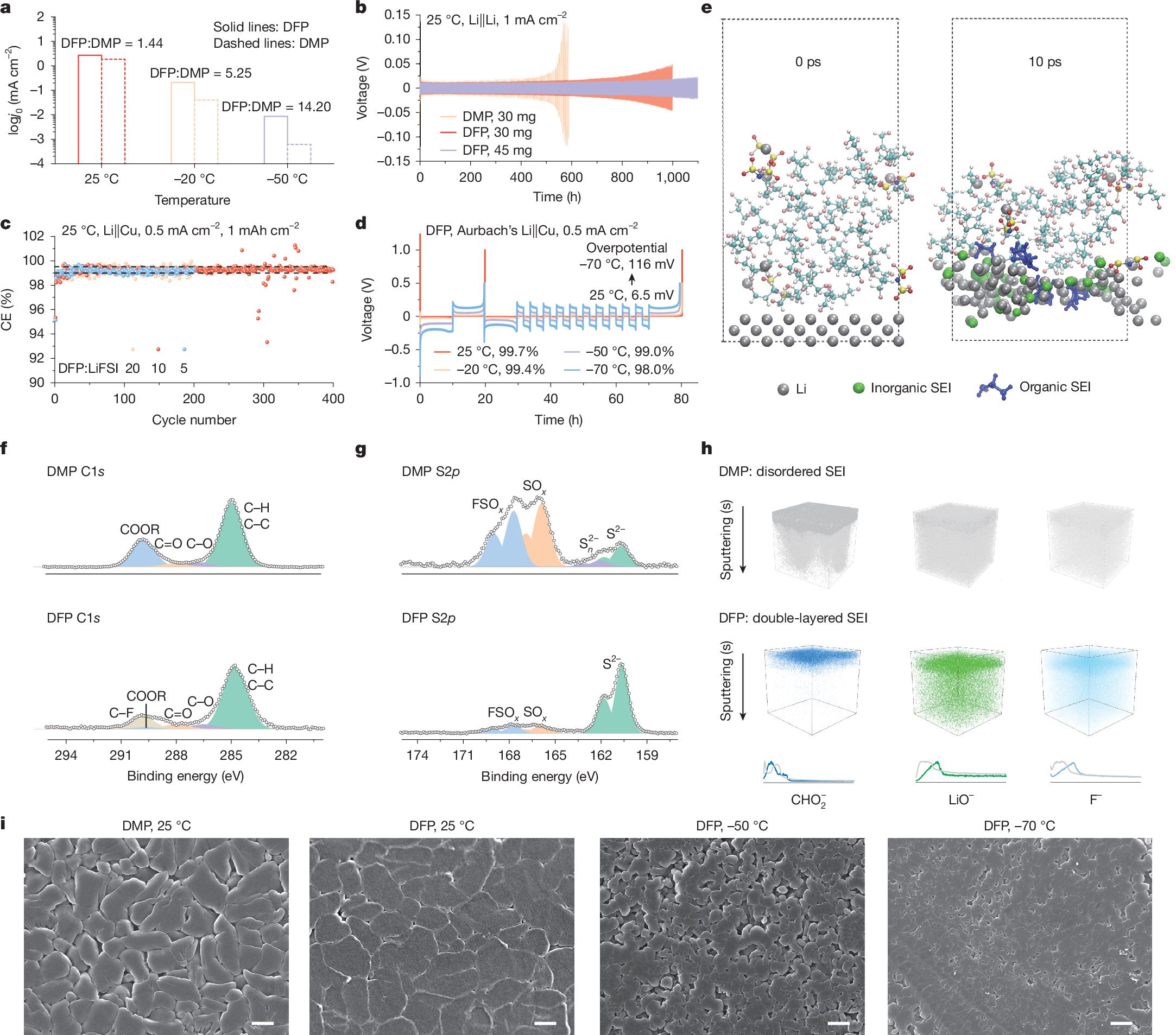
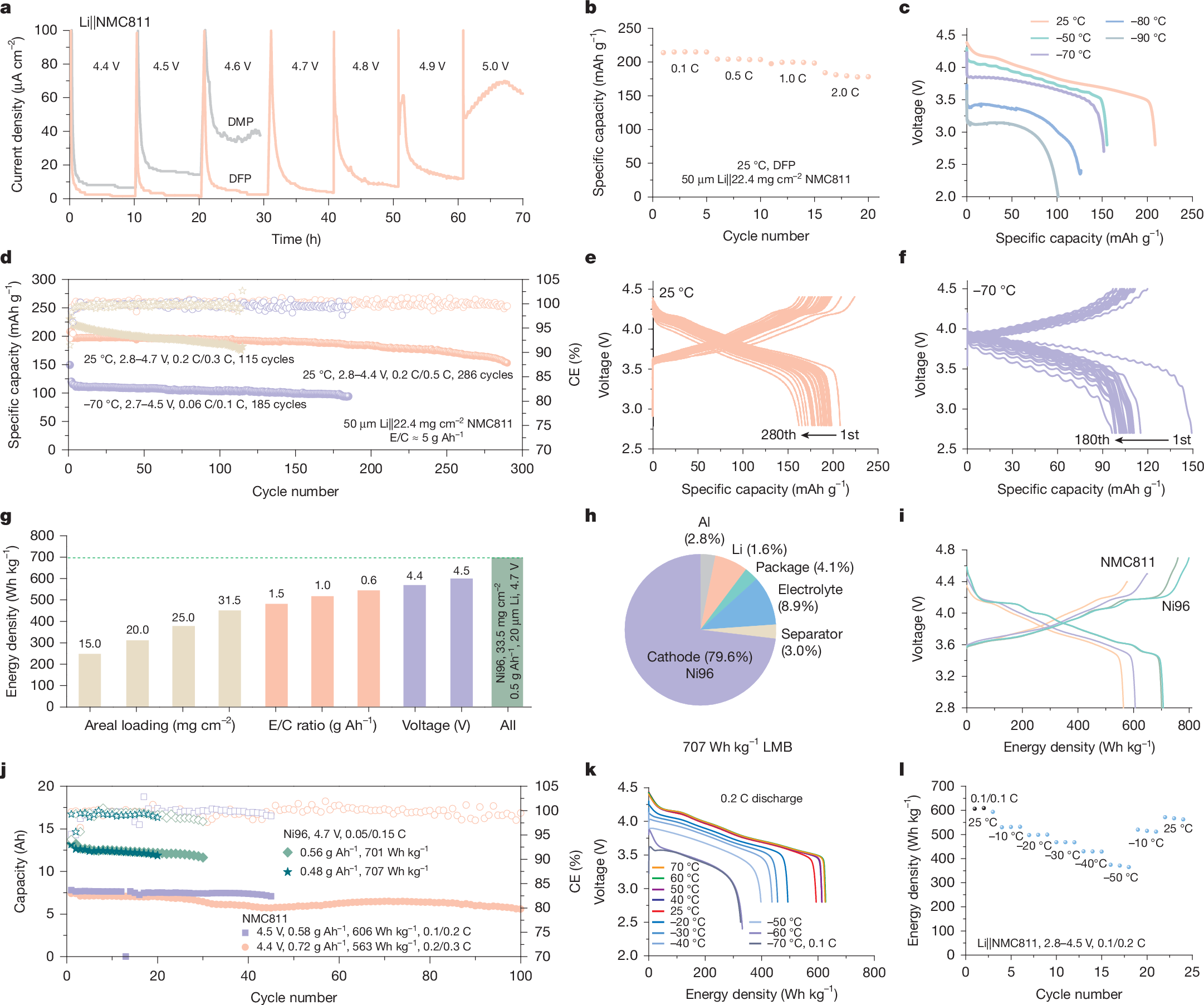
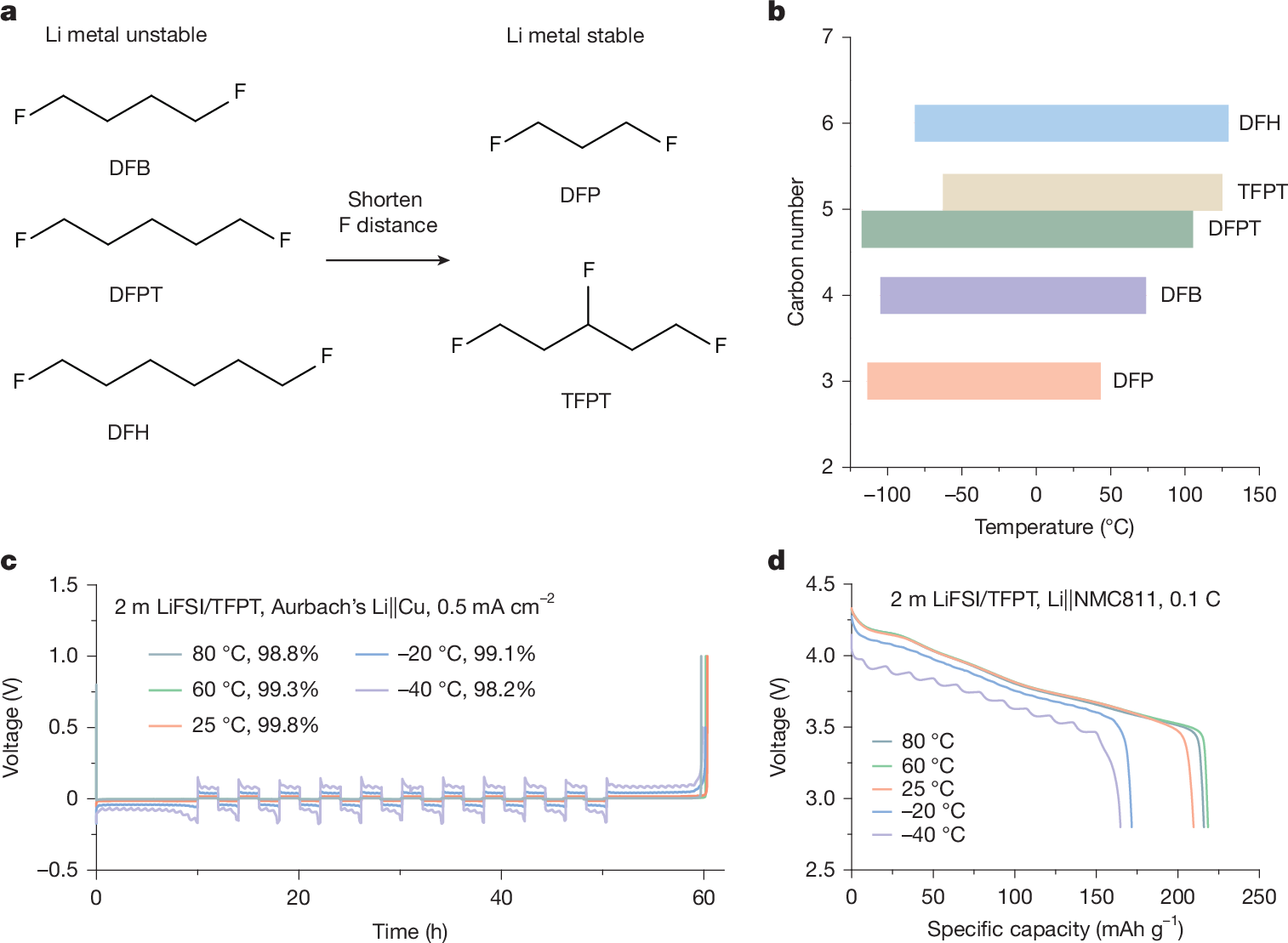
Electrolyte solvents for electrochemical devices have been dominated by oxygen (O)-based and nitrogen (N)-based ligands over the past decades1,2,3,4,5, for which the dipole–ion (Li+, Na+ and so on) interaction usually lays the foundations of ion dissociation and transport but frustrates the charge transfer process at the electrolyte–electrode interface6,7,8,9. Here, by synthesizing alkanes with monofluorinated structures, we show that fluorine (F)-based ligands with designed steric hindrance and Lewis basicity enable salt dissolution of more than 2 mol l−1. Among them, 1,3-difluoro-propane (DFP)-based Li-ion electrolyte is endowed with all merits for energy-dense and low-temperature batteries, including low viscosity (0.95 cp), high oxidation stability (>4.9 V) and ionic conductivity of 0.29 mS cm−1 at −70 °C. By incorporating F atoms in the first solvation shell, the weak F–Li+ coordination facilitates the Li plating/stripping process with Coulombic efficiency (CE) up to 99.7% and exchange current density one magnitude larger than O–Li+ coordination at −50 °C. The electrolytes further enable the operation of lithium-metal pouch cells under an electrolyte amount of less than 0.5 g Ah−1, achieving energy densities greater than 700 Wh kg−1 at room temperature and about 400 Wh kg−1 at −50 °C. The hydrofluorocarbon (HFC) electrolytes in this work provide a feasible approach to building electrochemical systems beyond traditional coordination chemistry.
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others

A monofluoride ether-based electrolyte solution for fast-charging and low-temperature non-aqueous lithium metal batteries
Rational solvent molecule tuning for high-performance lithium metal battery electrolytes

Flame-retardant electrolytes with electrochemically-inert and weakly coordinating dichloroalkane diluents for practical lithium metal batteries
Data availability
The data that support the findings of this study are available within this article and its Supplementary information. Further data are available from the corresponding authors on request. Source data are provided with this paper.
References
Lu, D. et al. Ligand-channel-enabled ultrafast Li-ion conduction. Nature 627, 101–107 (2024).
Fan, X. & Wang, C. High-voltage liquid electrolytes for Li batteries: progress and perspectives. Chem. Soc. Rev. 50, 10486–10566 (2021).
Jie, Y. et al. Towards long-life 500 Wh kg−1 lithium metal pouch cells via compact ion-pair aggregate electrolytes. Nat. Energy 9, 987–998 (2024).