Pancreatic-targeted lipid nanoparticles based on organ capsule filtration
TL;DR
Researchers developed a pancreatic-targeted lipid nanoparticle (AH-LNP) that selectively accumulates in the pancreas via capsule filtration and receptor-mediated endocytosis. This enables efficient mRNA delivery for genome editing and cancer therapy, showing promise in animal models and clinical translation.
Key Takeaways
- •AH-LNP uses size enlargement and capsule filtration for precise pancreatic targeting, enhancing mRNA delivery.
- •It enables effective genome editing in the pancreas for treating autoimmune diseases and improves antitumour efficacy in cancer models.
- •Safety and efficacy were validated in multiple animal models, including non-human primates, supporting potential clinical applications.
Tags
Abstract
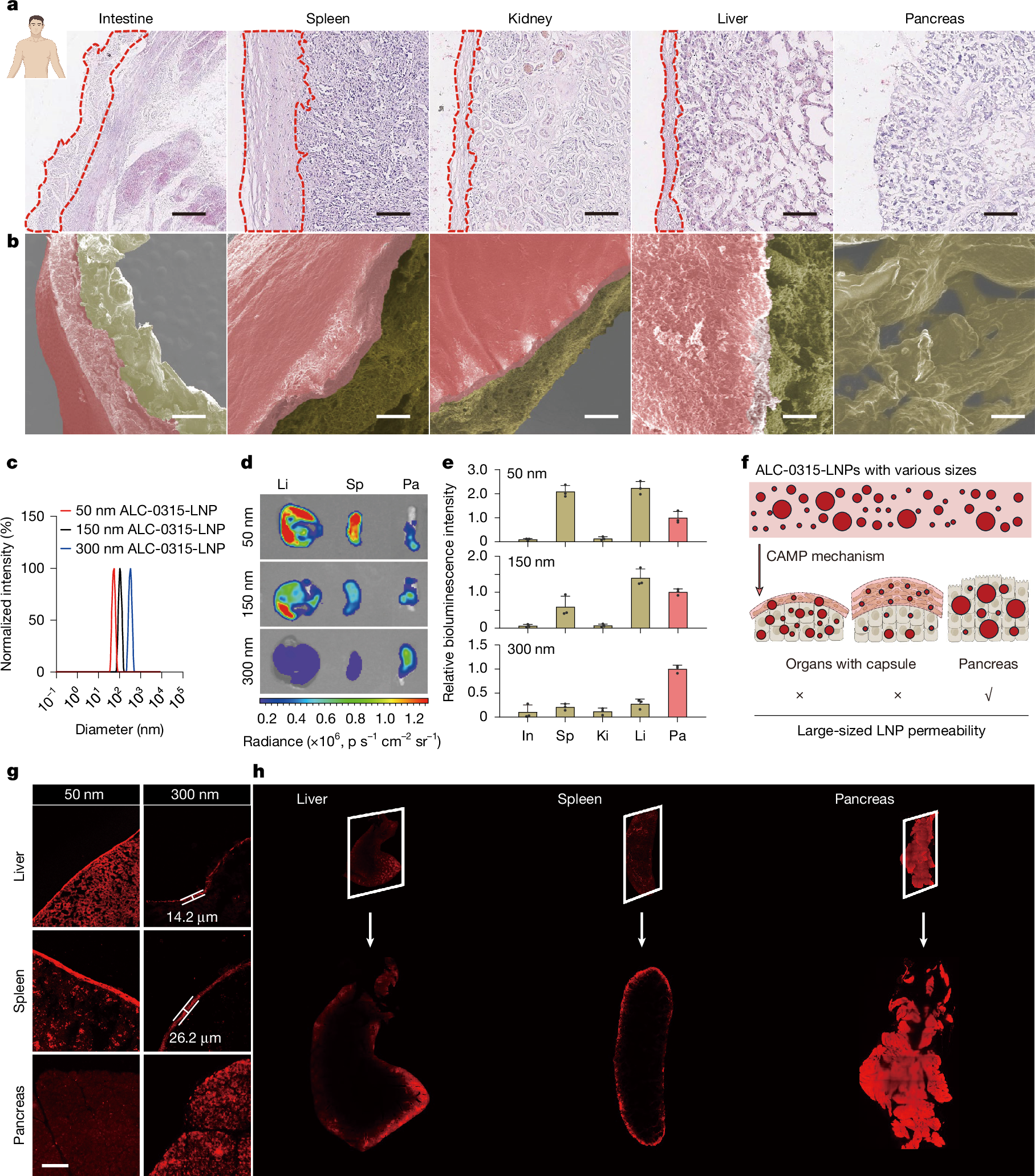
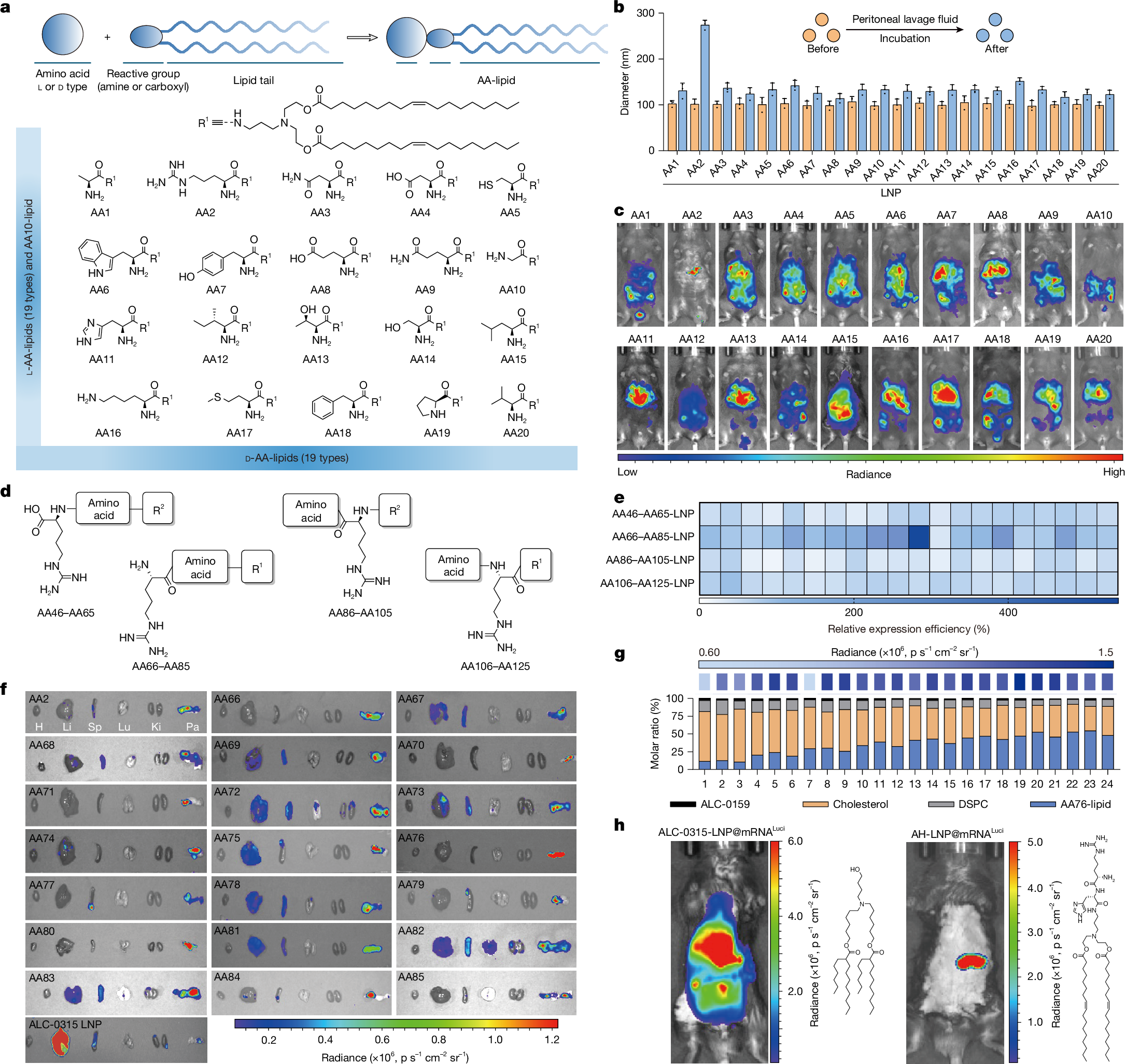
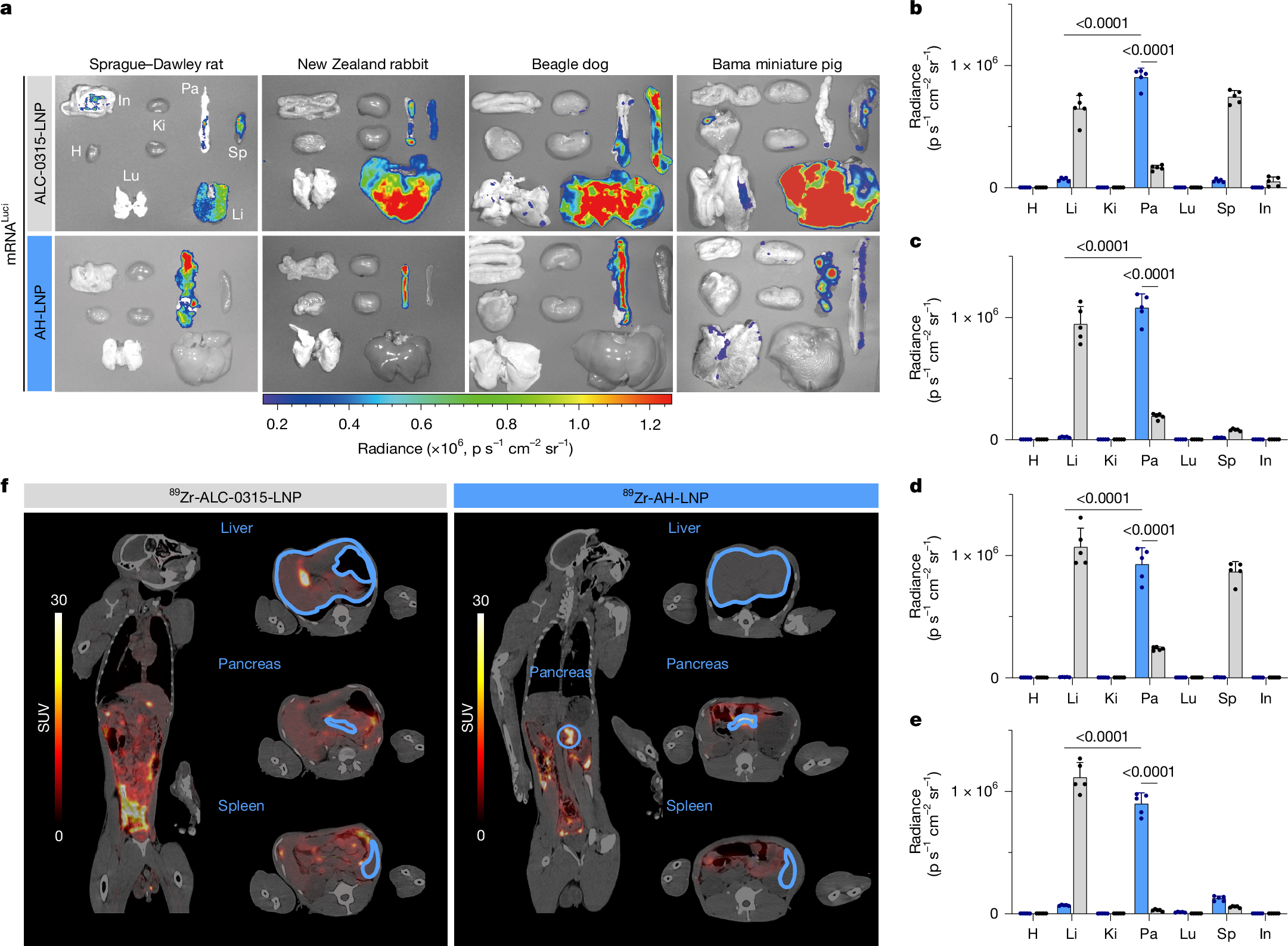
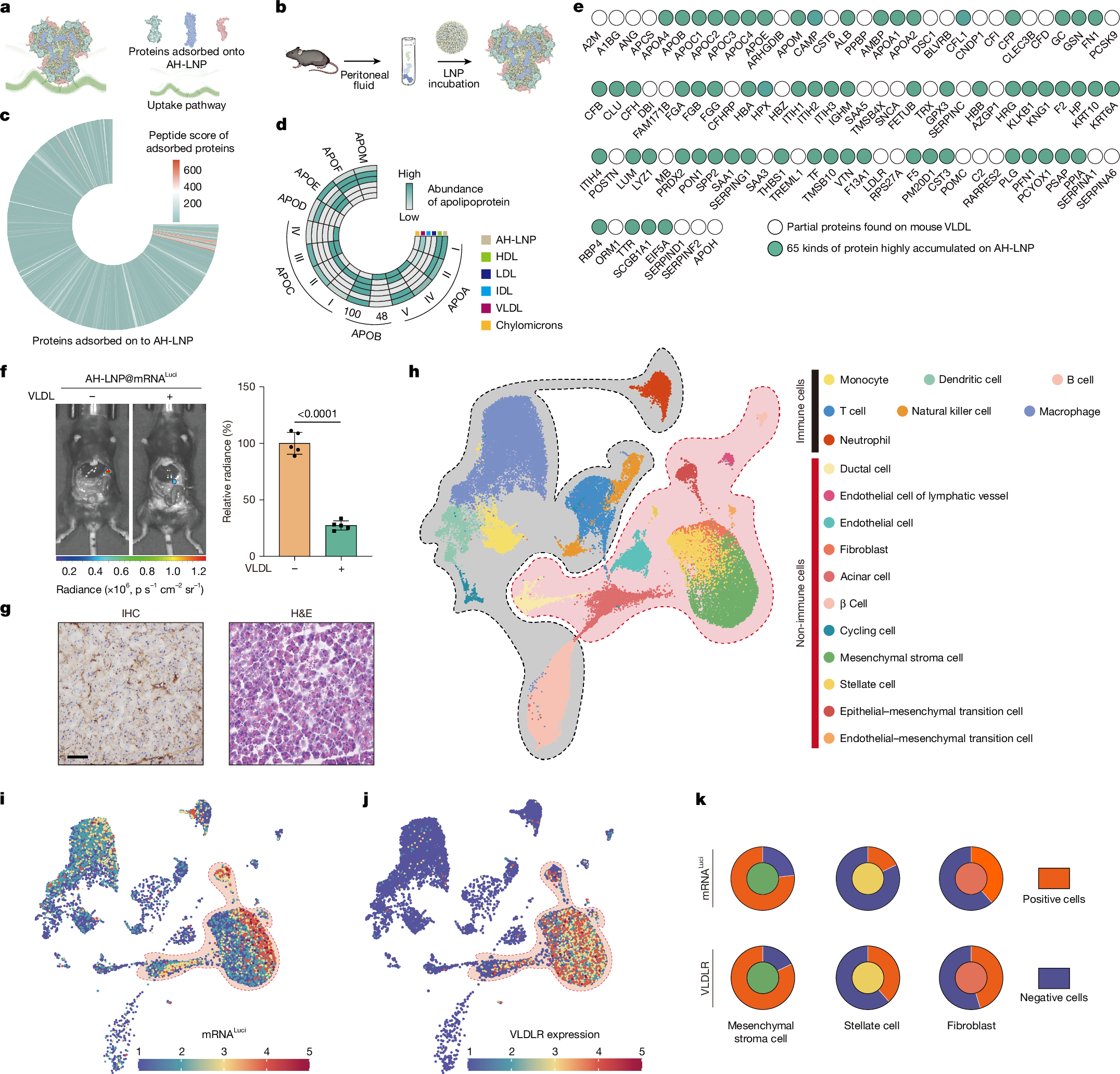
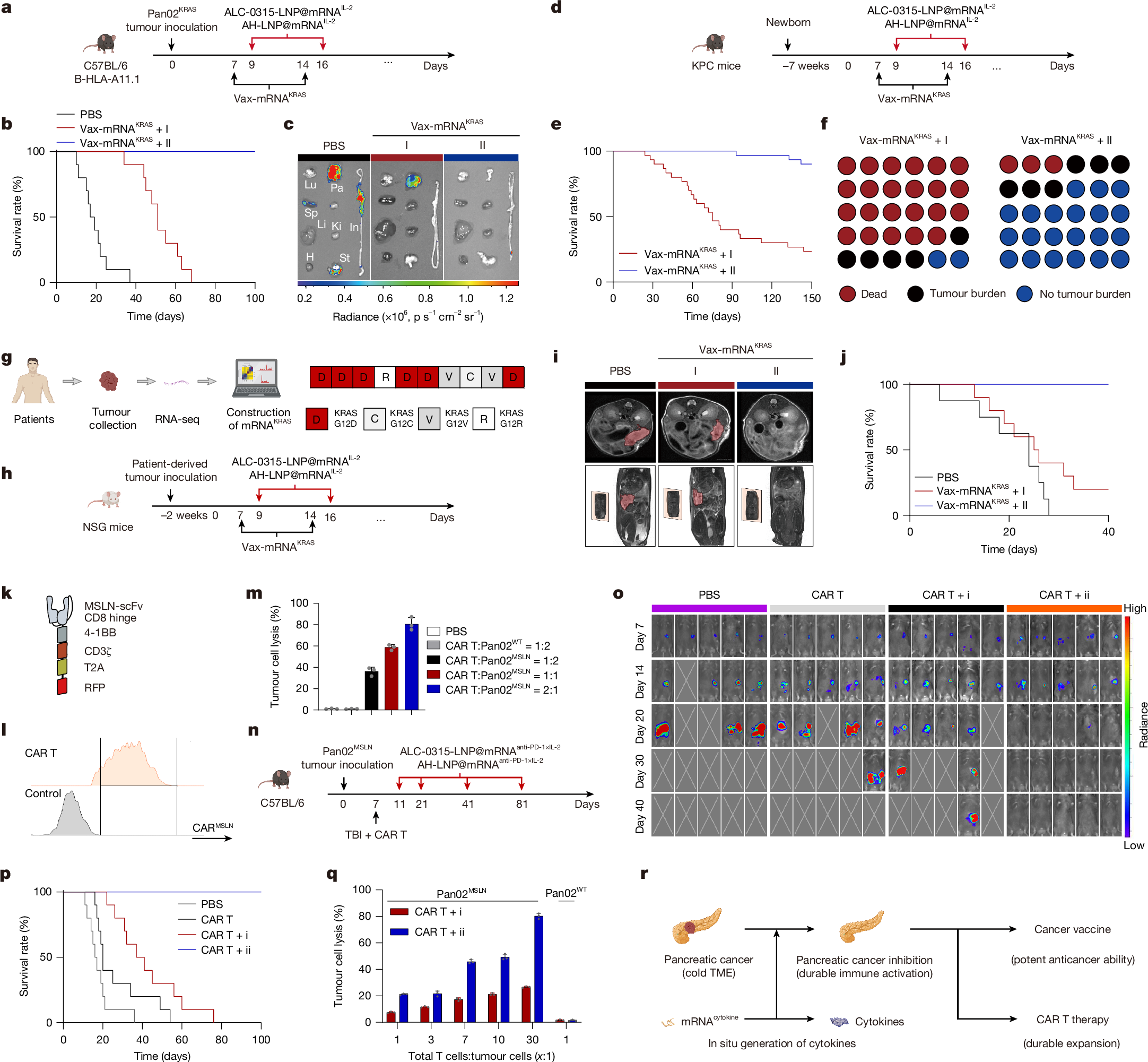
Achieving pancreatic-targeted delivery marks a breakthrough in treating pancreatic diseases, yet precise delivery remains challenging1. Here we identify an explicit and universal principle for pancreatic-selective delivery and propose a pancreatic-targeted lipid nanoparticle (AH-LNP) for mRNA delivery. AH-LNP exhibits size enlargement after assembly with proteins, facilitating capsule-filter-mediated pancreas-selective accumulation and receptor-mediated endocytosis, thereby boosting the pancreatic-targeted ability. Benefiting from this, AH-LNP enables precise and efficient genome editing in the pancreas through the delivery of Cas9 mRNA and single guide RNA (sgRNA), exhibiting promising potential in the treatment of autoimmune pancreatic diseases. Furthermore, pancreatic-targeted delivery of mRNA encoding therapeutic cytokines through AH-LNP demonstrates superior antitumour efficacy when combined with a cancer vaccine or chimeric antigen receptor T cell therapy in multiple pancreatic cancer models. The safety and pancreatic mRNA delivery of AH-LNP were verified in multiple animal models, including non-human primates, demonstrating great promise for clinical translation. Our findings highlight the transformative potential of this pancreatic-targeted mechanism and the derived LNP platform, opening avenues for developing precision therapeutics against diverse pancreatic diseases.
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
Acute pancreatitis: mechanisms and therapeutic approaches
Lnc-PFAR facilitates autophagy and exacerbates pancreatic fibrosis by reducing pre-miR-141 maturation in chronic pancreatitis

Migrasome-related LncRNA features predict immune microenvironment and prognosis in pancreatic cancer
Data availability
All data generated and supporting the findings of this study are available within the Article and its Supplementary Information. The raw sequencing data reported in this study have been deposited at the Genome Sequence Archive under accession number PRJCA051840. Data for VLDLR expression across various organs in humans are accessible in the DISCO36. Data on PD-L1 expression in pancreatic tumours and its correlation with survival are available in the GEPIA37. Source data are provided with this paper.
References
GBD 2021 Diabetes Collaborators. Global, regional, and national burden of diabetes from 1990 to 2021, with projections of prevalence to 2050: a systematic analysis for the Global Burden of Disease Study 2021. Lancet 402, 203–234 (2023).
Siegel, R. L. et al. Cancer statistics, 2023. CA Cancer J. Clin. 73, 17–48 (2023).
Barbier, A. J., Jiang, A. Y., Zhang, P., Wooster, R. & Anderson, D. G. The clinical progress of mRNA vaccines and immunotherapies. Nat. Biotechnol. 40, 840–854 (2022).
Dilliard, S. A. & Siegwart, D. J. Passive, active and endogenous organ-targeted lipid and polymer nanoparticles for delivery of genetic drugs. Nat. Rev. Mater. 8, 282–300 (2023).