A metabolic alarmin from keratinocytes potentiates systemic humoral immunity
TL;DR
Farnesyl pyrophosphate (FPP) from keratinocytes acts as an alarmin, enhancing systemic antibody responses via the TRPV3–IL-6/CCL20–germinal center axis. This pathway is activated by infection or UV irradiation and contributes to both protective immunity and disease pathology in conditions like SLE.
Key Takeaways
- •FPP, a metabolic intermediate, functions as an endogenous alarmin that boosts IgG antibody production through keratinocyte-derived IL-6 and CCL20.
- •The FPP–TRPV3–IL-6/CCL20 axis promotes T follicular helper cell differentiation and germinal center responses, enhancing pathogen-specific immunity.
- •Hyperactivation of this signaling pathway in skin lesions correlates with exacerbated pathology in systemic lupus erythematosus (SLE), suggesting therapeutic targets.
Tags
Abstract
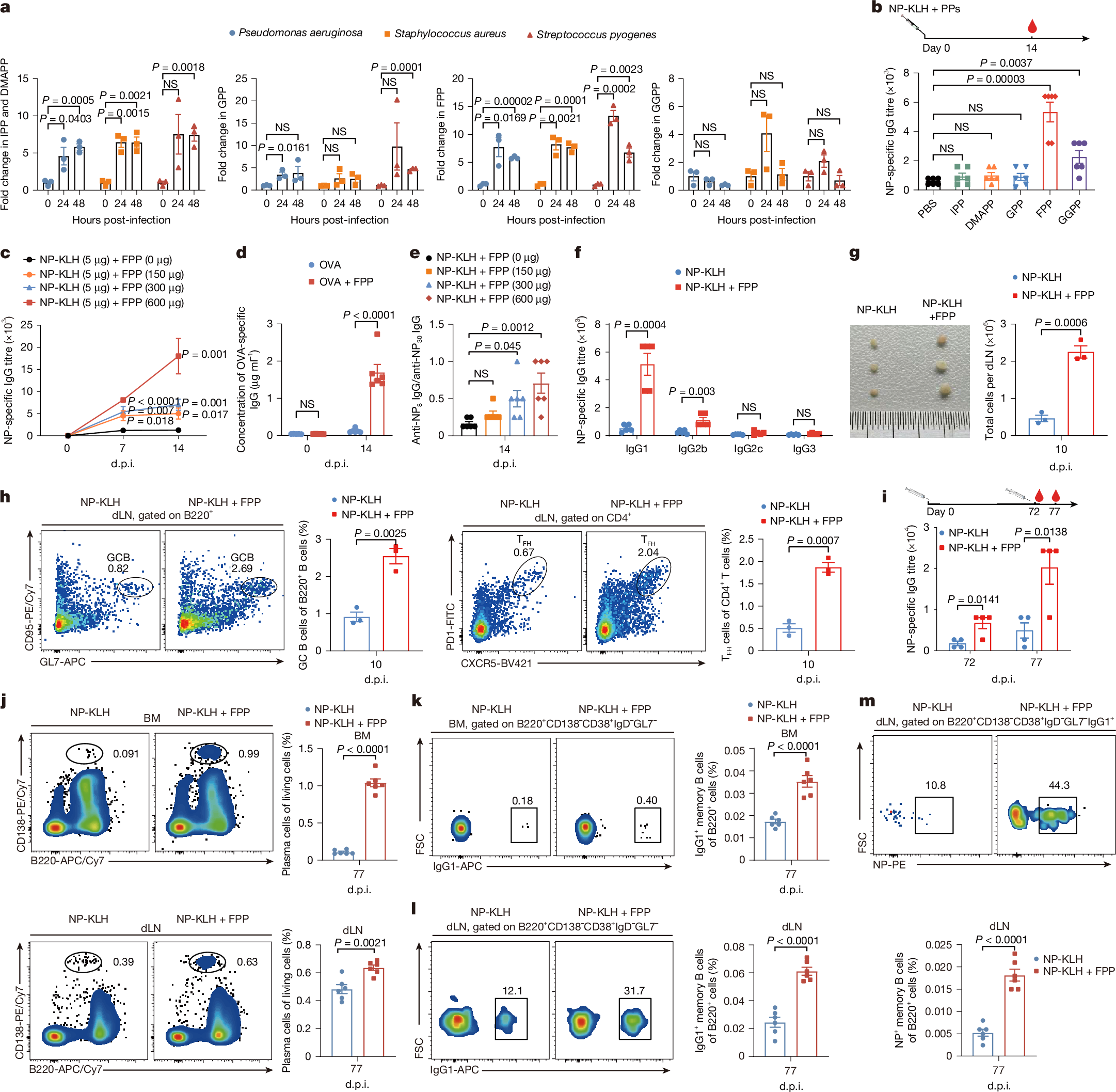
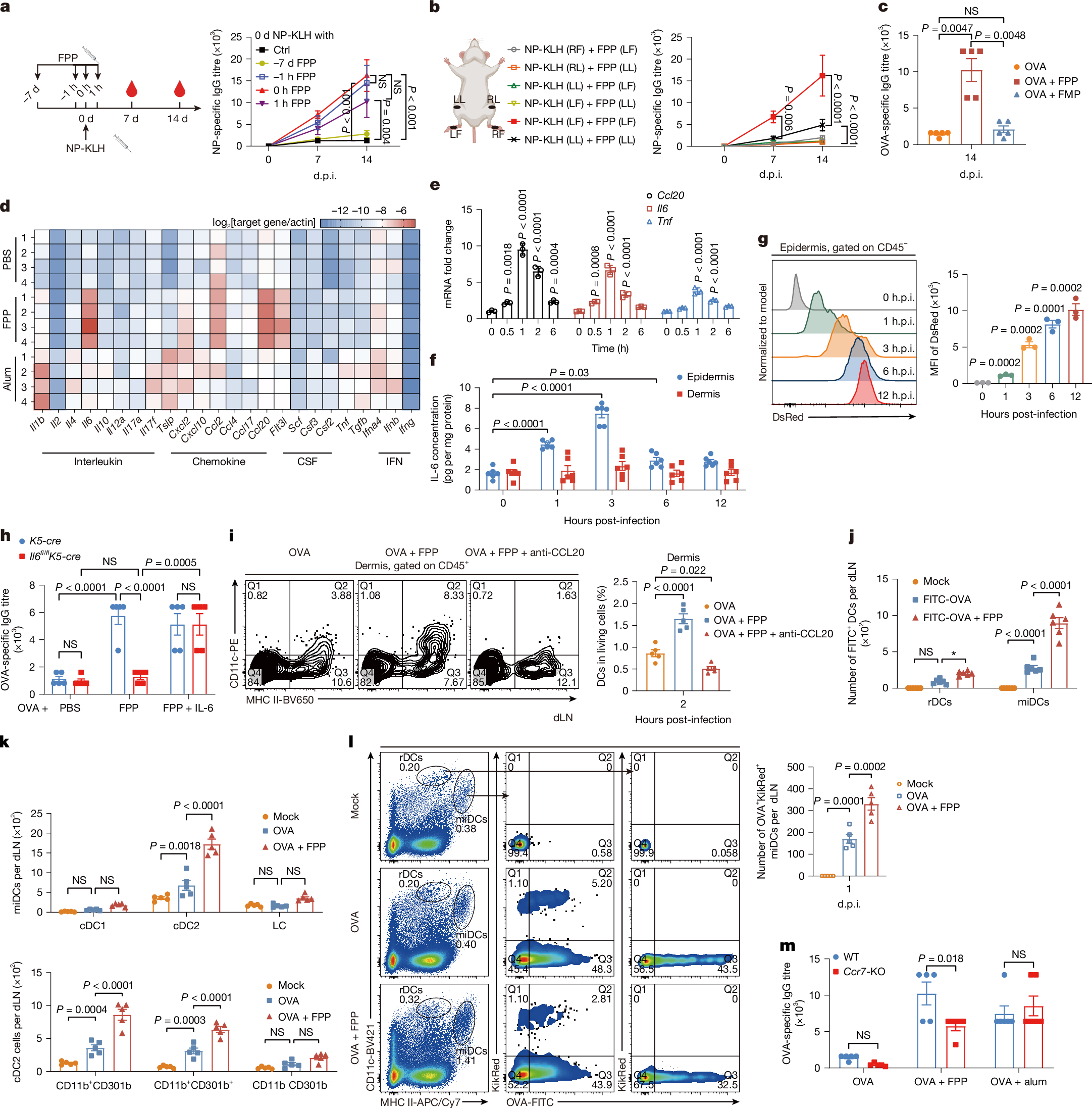
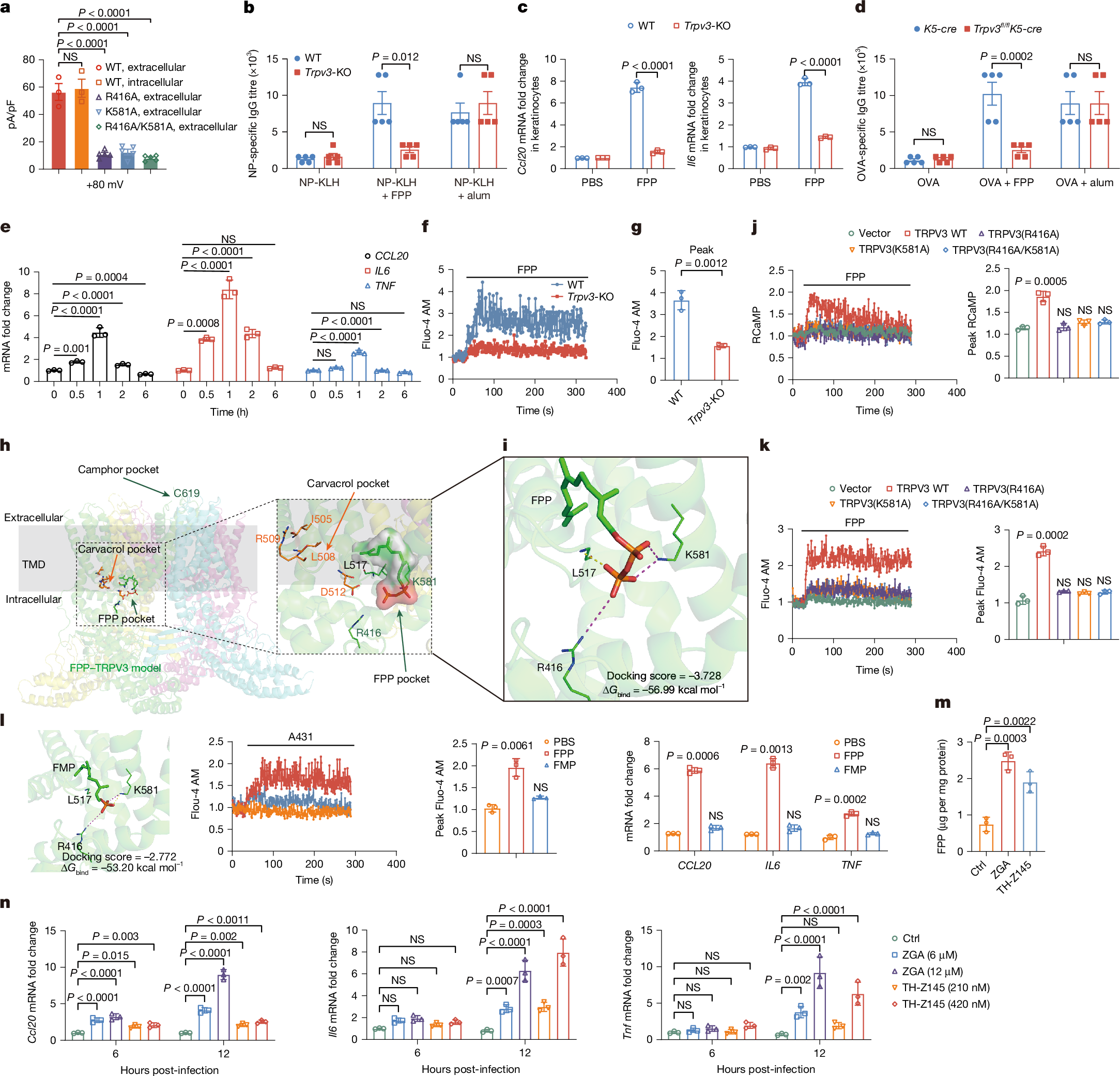
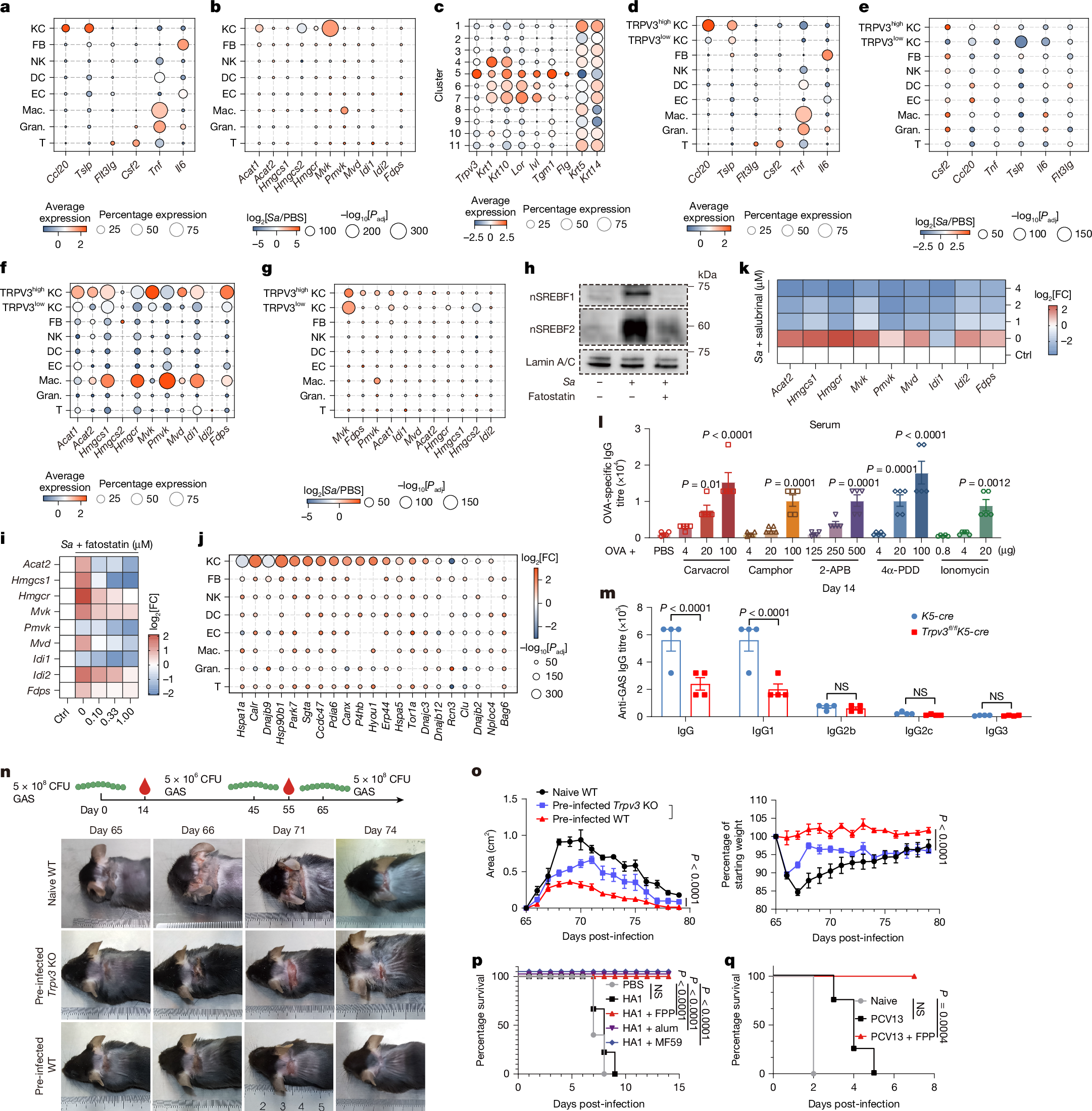
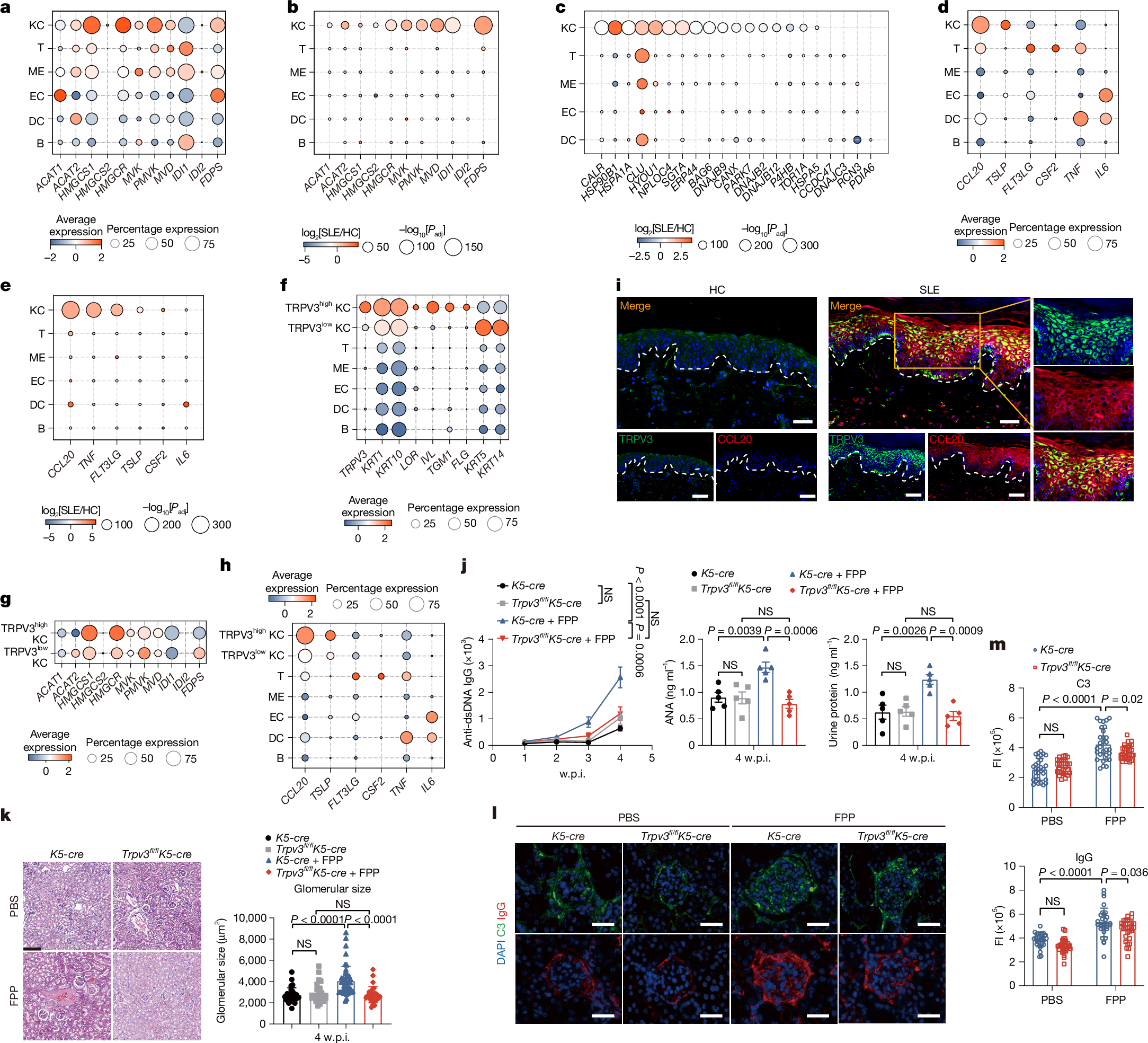
How a local infection triggers systemic humoral immunity remains unclear. Here we identify farnesyl pyrophosphate (FPP), a mevalonate pathway metabolic intermediate1, as an endogenous alarmin that enhances IgG antibody responses through keratinocyte-derived IL-6 and CCL20. This signalling axis potentiates the differentiation of T follicular helper cells and migratory dendritic cells2,3. FPP accumulates within keratinocytes after infection or ultraviolet irradiation through the activation of the mevalonate pathway mediated by the unfolded protein response–SREBF pathway, amplifying germinal centre (GC) responses in draining lymph nodes. Mechanistically, accumulated FPP in the cytosol engages transient receptor potential vanilloid 3 (TRPV3) by binding to its intracellular domains, inducing Ca2+ influx that subsequently activates the calmodulin–calcineurin–NFAT and PYK2–RAS–ERK pathways to enhance IL-6 and CCL20 production. This FPP–TRPV3–IL-6/CCL20–GC axis potentiates pathogen-specific antibody production, conferring protection in wild-type but not TRPV3-deficient mice. Single-cell RNA-sequencing analyses of systemic lupus erythematosus (SLE) skin lesions and pathogen-infected mouse skin demonstrate hyperactivation of this signalling axis, particularly in the TRPV3high keratinocyte subset. In mouse models of SLE, the activation of this axis correlates with exacerbated disease pathology. Thus, FPP potentiates systemic humoral immunity through the TRPV3–IL-6/CCL20–GC signalling axis, providing insights for the development of vaccine adjuvants and potential therapeutics for SLE.
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout
Data availability
For the S. aureus mouse skin infection dataset, we downloaded publicly available scRNA-seq data from the SRA under accession number PRJNA975496. For the SLE and healthy control epidermis datasets, we downloaded publicly available scRNA-seq data from the GEO under accession numbers GSE179633 and GSE281449. For the healthy volunteer skin scATAC–seq dataset, we downloaded publicly available data from the GEO under accession number GSE212450. For the UVB-treated healthy volunteer skin bulk RNA-seq dataset, we downloaded publicly available data from the GEO under accession number GSE148535. The baseline expression data of TRPV3, TRPM2 and HMGCR in human tissue, cell types and cell lines were obtained from Human Protein Atlas (www.proteinatlas.org)94,95. Further information and requests for resources and reagents should first be directed to W.L. Source data are provided with this paper.
References
Lange, B. M. et al. Isoprenoid biosynthesis: the evolution of two ancient and distinct pathways across genomes. Proc. Natl Acad. Sci. USA 97, 13172–13177 (2000).
Dienz, O. et al. The induction of antibody production by IL-6 is indirectly mediated by IL-21 produced by CD4+ T cells. J. Exp. Med. 206, 69–78 (2009).
Sun, X. et al. Membrane-anchored CCL20 augments HIV Env-specific mucosal immune responses. Virol. J. 14, 163 (2017).
Pasparakis, M. et al. Mechanisms regulating skin immunity and inflammation. Nat. Rev. Immunol. 14, 289–301 (2014).
Kashem, S. W. et al. Antigen-presenting cells in the skin. Annu. Rev. Immunol. 35, 469–499 (2017).
Kabashima, K. et al. The immunological anatomy of the skin. Nat. Rev. Immunol. 19, 19–30 (2019).