Lipid metabolism drives dietary effects on T cell ferroptosis and immunity
TL;DR
Dietary fat composition, particularly PUFA/MUFA ratios, regulates T cell ferroptosis resistance through lipid metabolism. This affects T cell homeostasis, humoral immunity, and anti-tumor responses, including CAR-T therapy. Targeting lipid metabolism could enhance immunotherapy efficacy.
Key Takeaways
- •Dietary fatty acid composition (PUFAs vs MUFAs) directly influences T cell resistance to ferroptosis in both mice and humans
- •Lipid metabolism drives dietary effects on T cell function, impacting follicular helper T cell generation and anti-tumor immunity
- •Ferroptosis regulation in T cells affects humoral immunity and CAR-T therapy outcomes
- •ACSL4 enzyme plays a key mechanistic role in linking dietary lipids to T cell ferroptosis regulation
- •Plasma lipid profiles correlate with human T cell ferroptosis resistance across healthy cohorts
Tags
Abstract
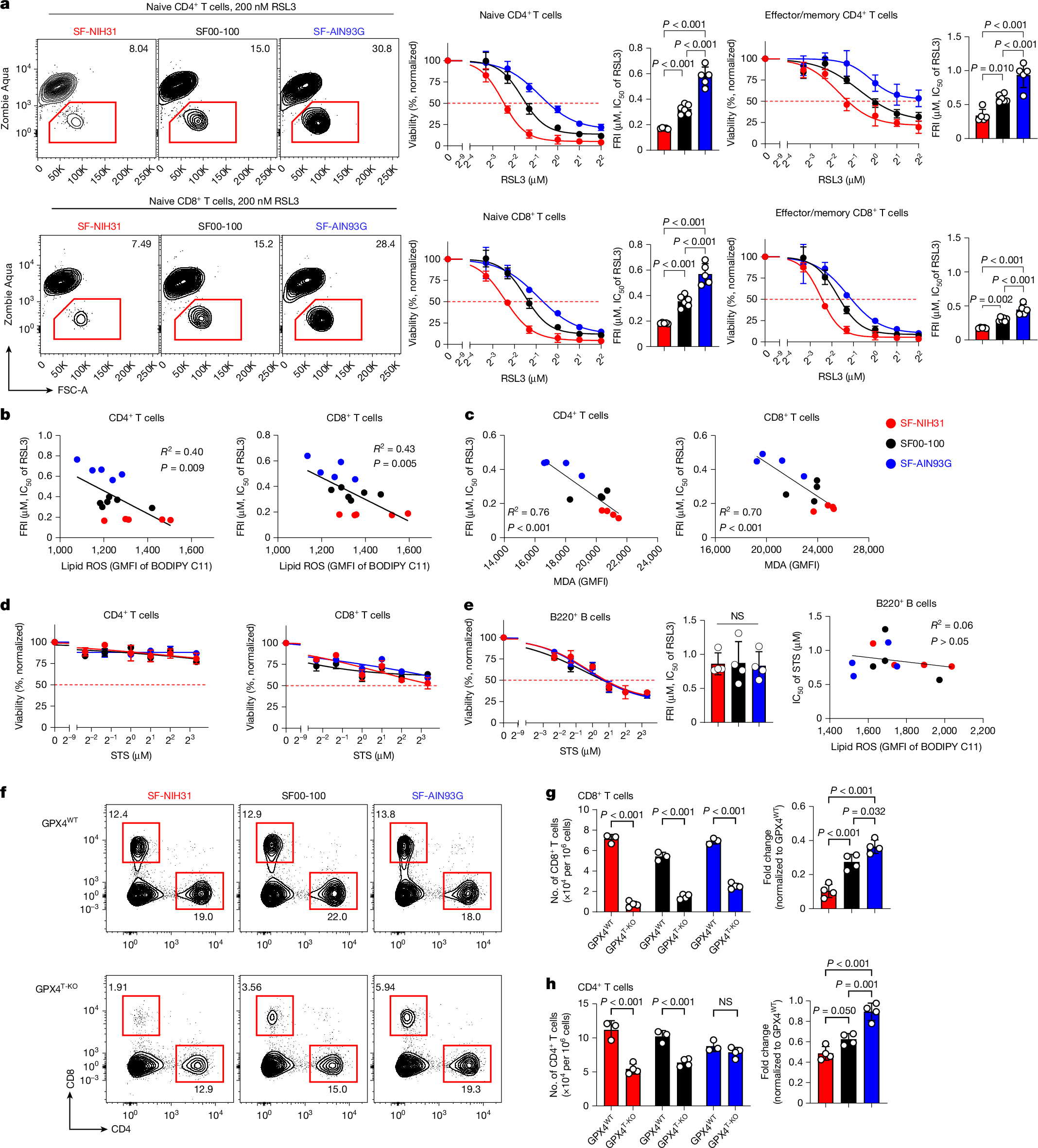
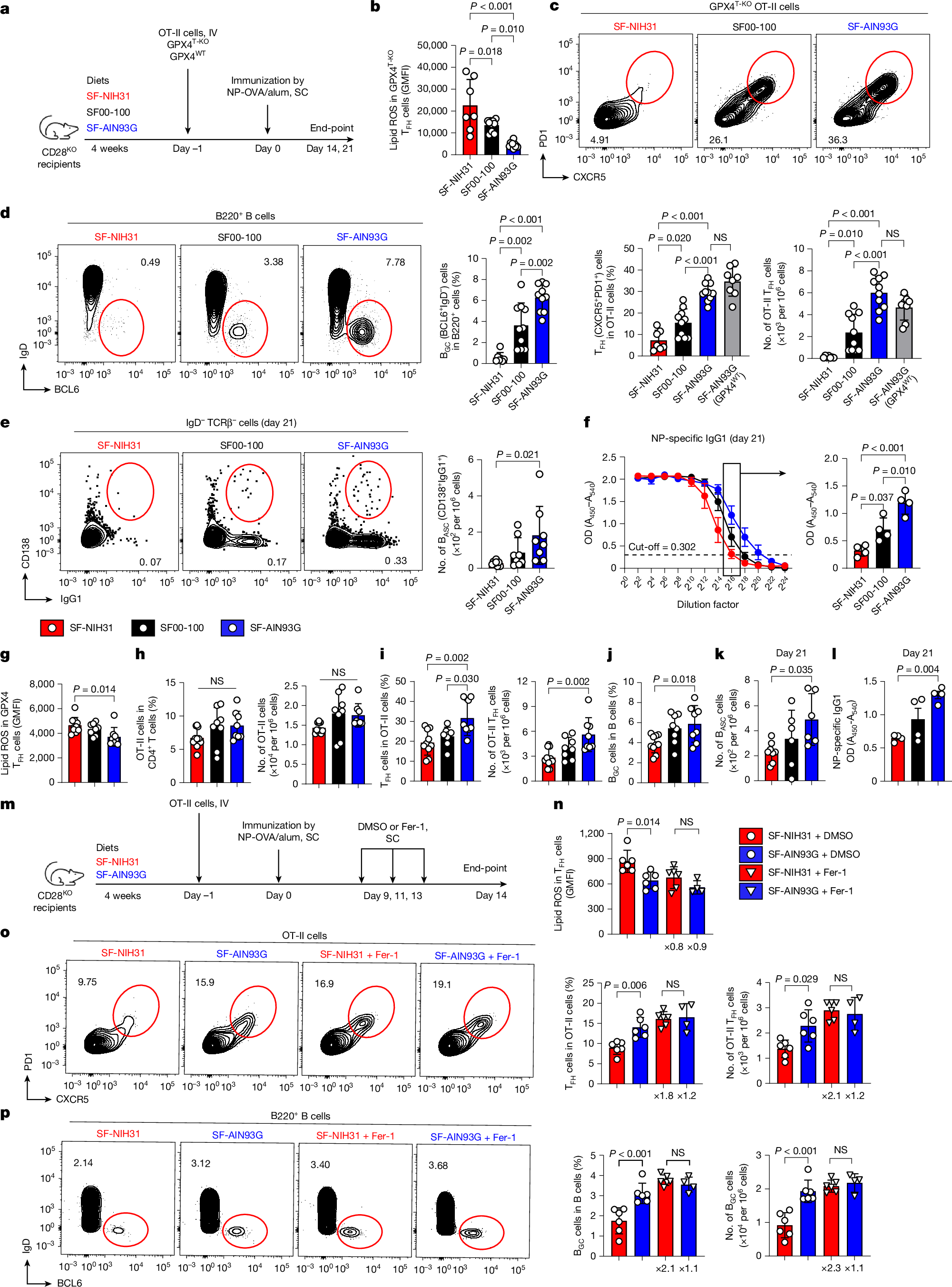
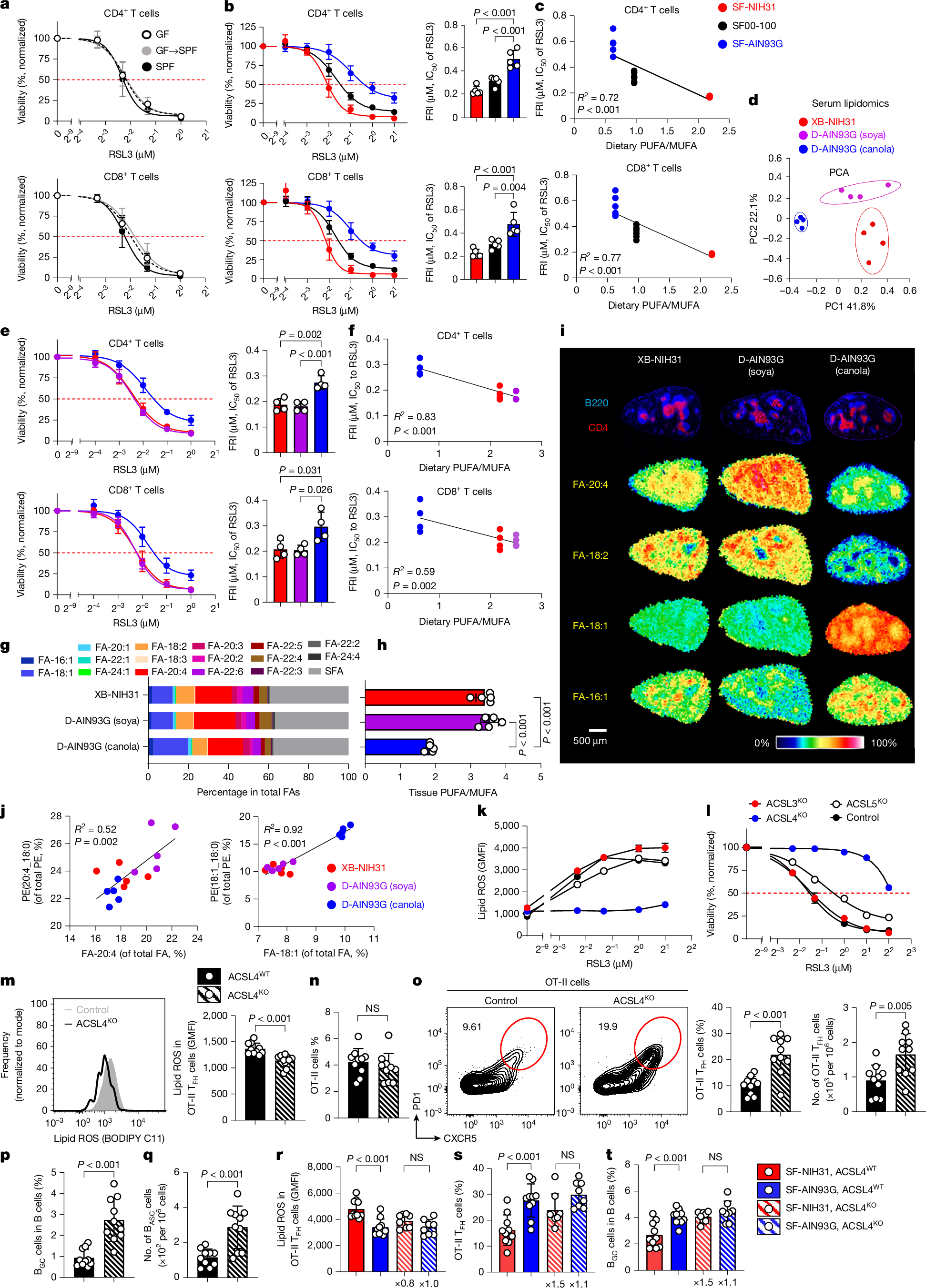
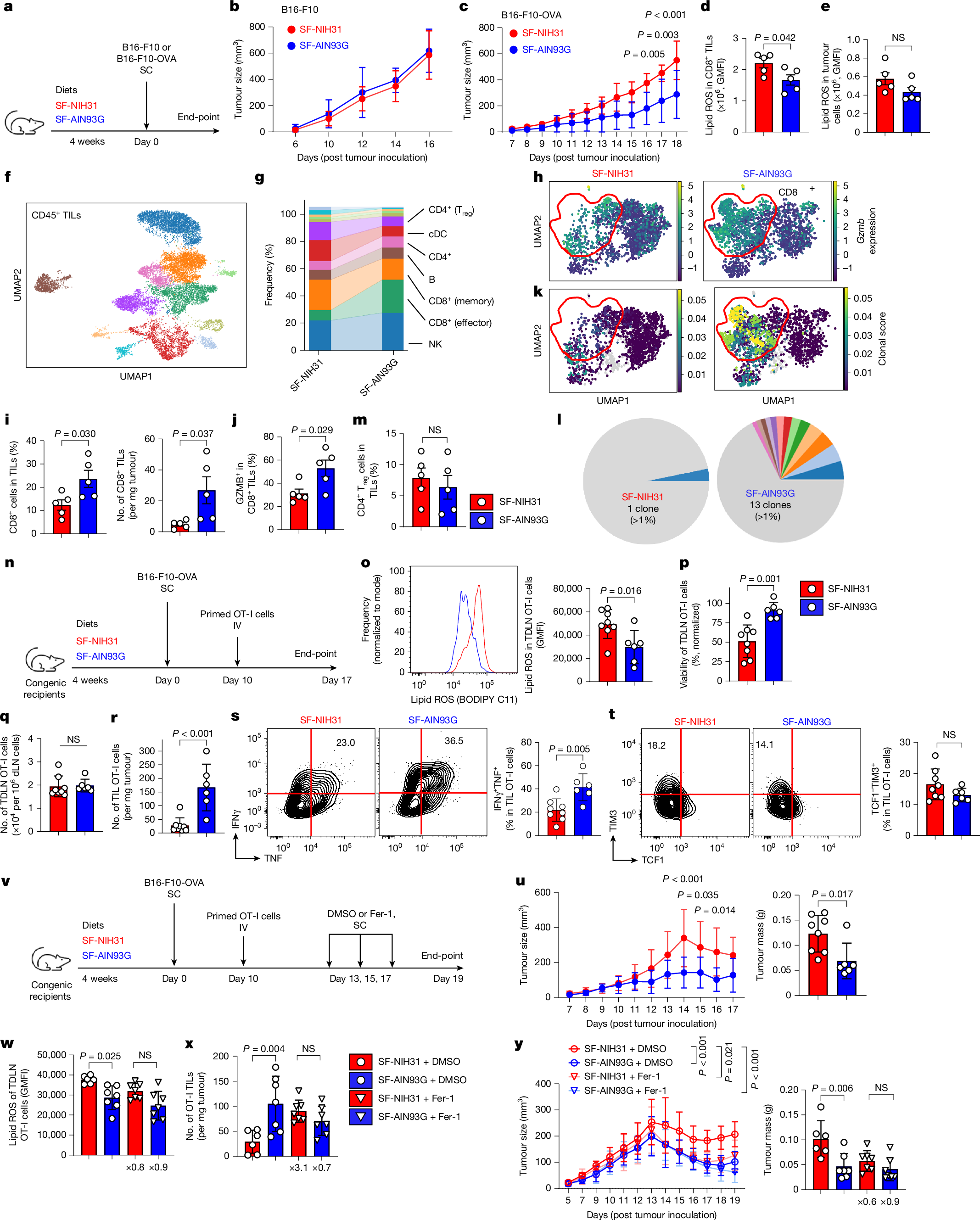
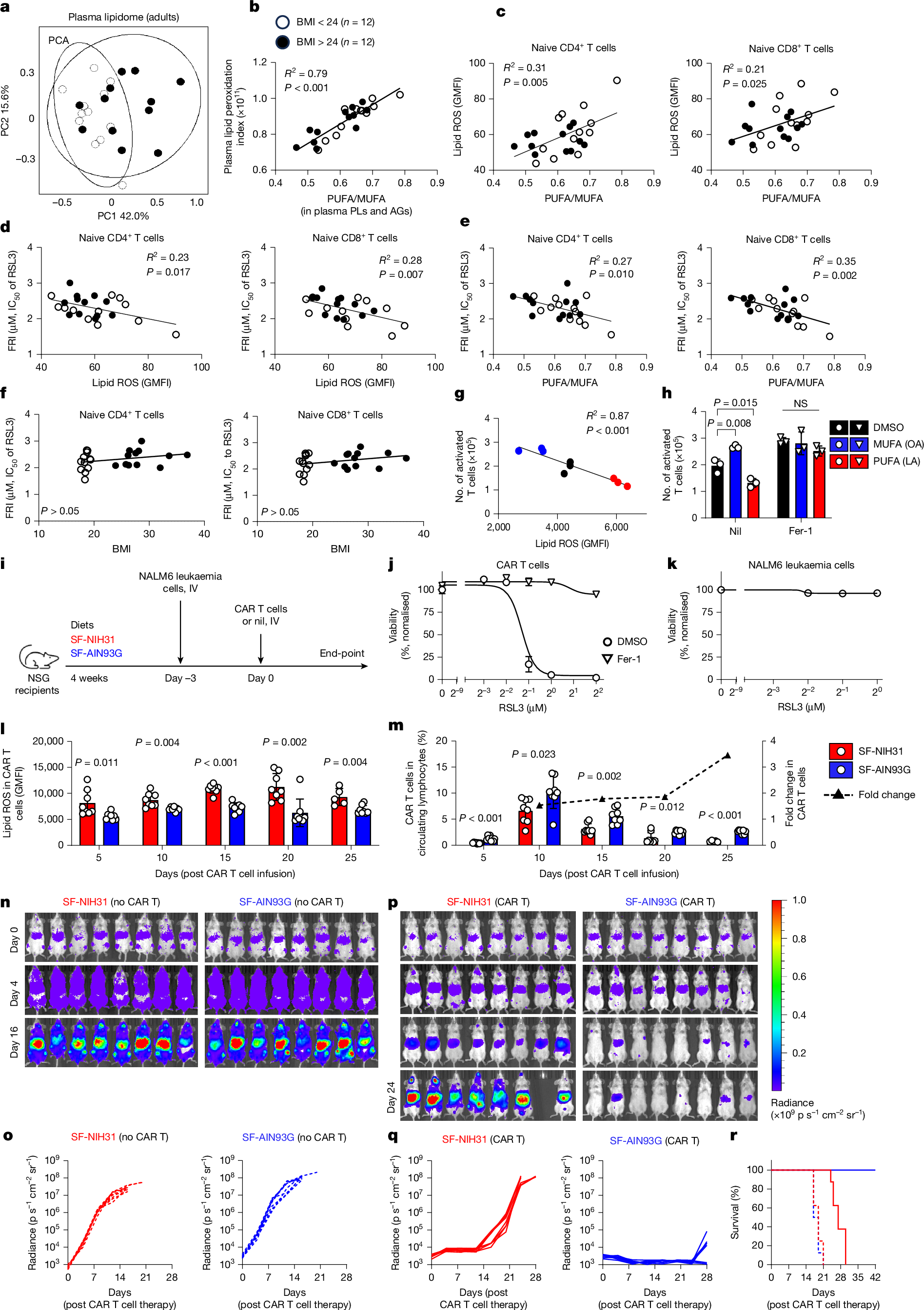
Ferroptosis, a major mechanism of non-apoptotic programmed cell death, critically regulates the homeostasis and functionality of peripheral CD4+ and CD8+ T cells1,2,3,4,5,6. Here we demonstrate that in mouse, resistance of T cells to ferroptosis depends critically on the composition of standard rodent diets, and that dietary effects on ferroptosis (DEFs) have a crucial role in regulation of T cell homeostasis and immune responses. DEFs are microbiota-independent and are driven by variations in dietary polyunsaturated and monounsaturated fatty acids (PUFAs and MUFAs) that lead to variations in abundance of lipid species in lymphoid tissues and T cells. Consistently, ferroptosis resistance of human T cells also correlated with plasma lipid profiles across multiple healthy cohorts, exhibiting negative associations with PUFA/MUFA ratios in major lipid classes. DEFs dictate T cell resilience in the absence of the essential lipid peroxide scavenger GPX4 and broadly modulate T cell-dependent humoral immunity and T cell-mediated anti-tumour immunity, including in chimeric antigen receptor T cell therapy. Mechanistically, ACSL4, which preferentially biosynthezises PUFA-containing phospholipids7, is highly expressed in T cells and underpins DEF-mediated regulation of follicular helper T (TFH) cell generation and function. Our findings reveal the physiological significance of lipid metabolism in driving DEFs in immunity and suggest strategies targeting lipid metabolism to enhance vaccine efficacy and T cell-mediated immunotherapy.
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout
Data availability
All of the data supporting the findings of this study are available from the corresponding author. RNA-sequencing and scRNA-seq data described in the article have been deposited in the EMBL-EBI database and are accessible via accession number E-MTAB-16107 (bulk RNA-seq) and E-MTAB-16518 (scRNA-seq). Publicly available normalized and batch-adjusted RNA-sequencing data were obtained from GSE137122 (mouse TFH cells and non-TFH cells post-immunization). Source data are provided with this paper.
Code availability
No new algorithms were developed for this Article. Processed data and analysis code are available from the corresponding author.
References
Matsushita, M. et al. T cell lipid peroxidation induces ferroptosis and prevents immunity to infection. J. Exp. Med. 212, 555–568 (2015).
Yao, Y. et al. Selenium–GPX4 axis protects follicular helper T cells from ferroptosis. Nat. Immunol. 22, 1127–1139 (2021).
Xu, C. et al. The glutathione peroxidase Gpx4 prevents lipid peroxidation and ferroptosis to sustain Treg cell activation and suppression of antitumor immunity. Cell Rep. 35, 109235 (2021).
Ma, X. et al. CD36-mediated ferroptosis dampens intratumoral CD8+ T cell effector function and impairs their antitumor ability. Cell Metab. 33, 1001–1012.e5 (2021).
Wang, Y. et al. The kinase complex mTORC2 promotes the longevity of virus-specific memory CD4+ T cells by preventing ferroptosis. Nat. Immunol. 23, 303–317 (2022).
Xu, S. et al. Uptake of oxidized lipids by the scavenger receptor CD36 promotes lipid peroxidation and dysfunction in CD8+ T cells in tumors. Immunity 54, 1561–1577.e1567 (2021).