Long-term thrombus-free left atrial appendage occlusion via magnetofluids
TL;DR
Researchers propose a new left atrial appendage occlusion method using magnetofluids that form magnetogels in the LAA. This approach shows long-term thrombus-free occlusion in animal models without device-related complications seen with traditional occluders.
Key Takeaways
- •Magnetofluids injected into the left atrial appendage can form magnetogels that provide complete, personalized occlusion
- •In pig models, the magnetogel showed long-term resilience and biocompatibility for over 10 months without thrombus formation
- •The magnetogel created a smooth, firm endocardium without crevices or myocardial injury, unlike traditional Watchman occluders
- •This approach addresses key limitations of current LAAO devices including peri-device leak and device-related thrombus
- •The method is suitable for all LAA types and may offer a promising clinical strategy for stroke prevention in atrial fibrillation patients
Tags
Abstract
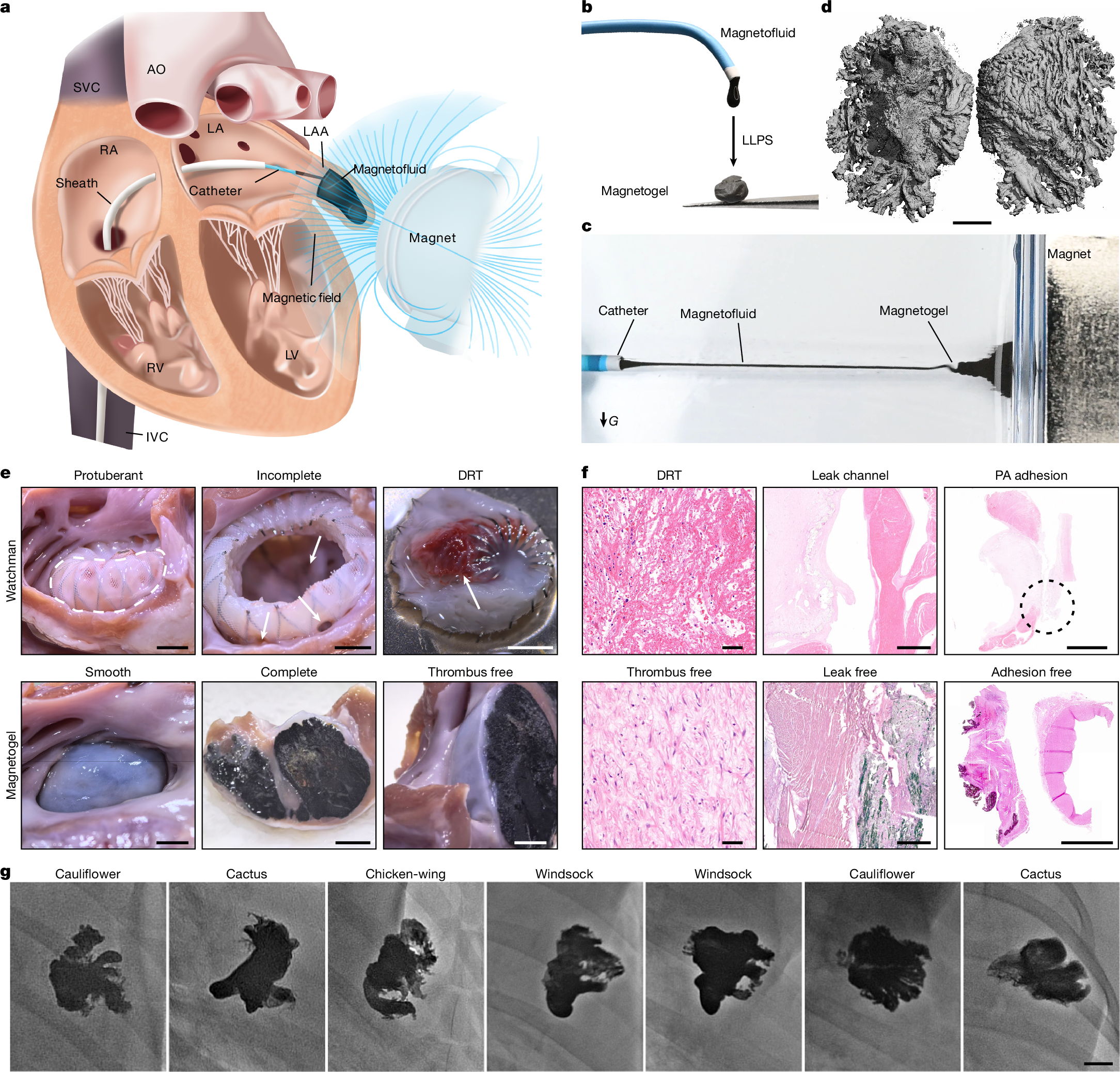
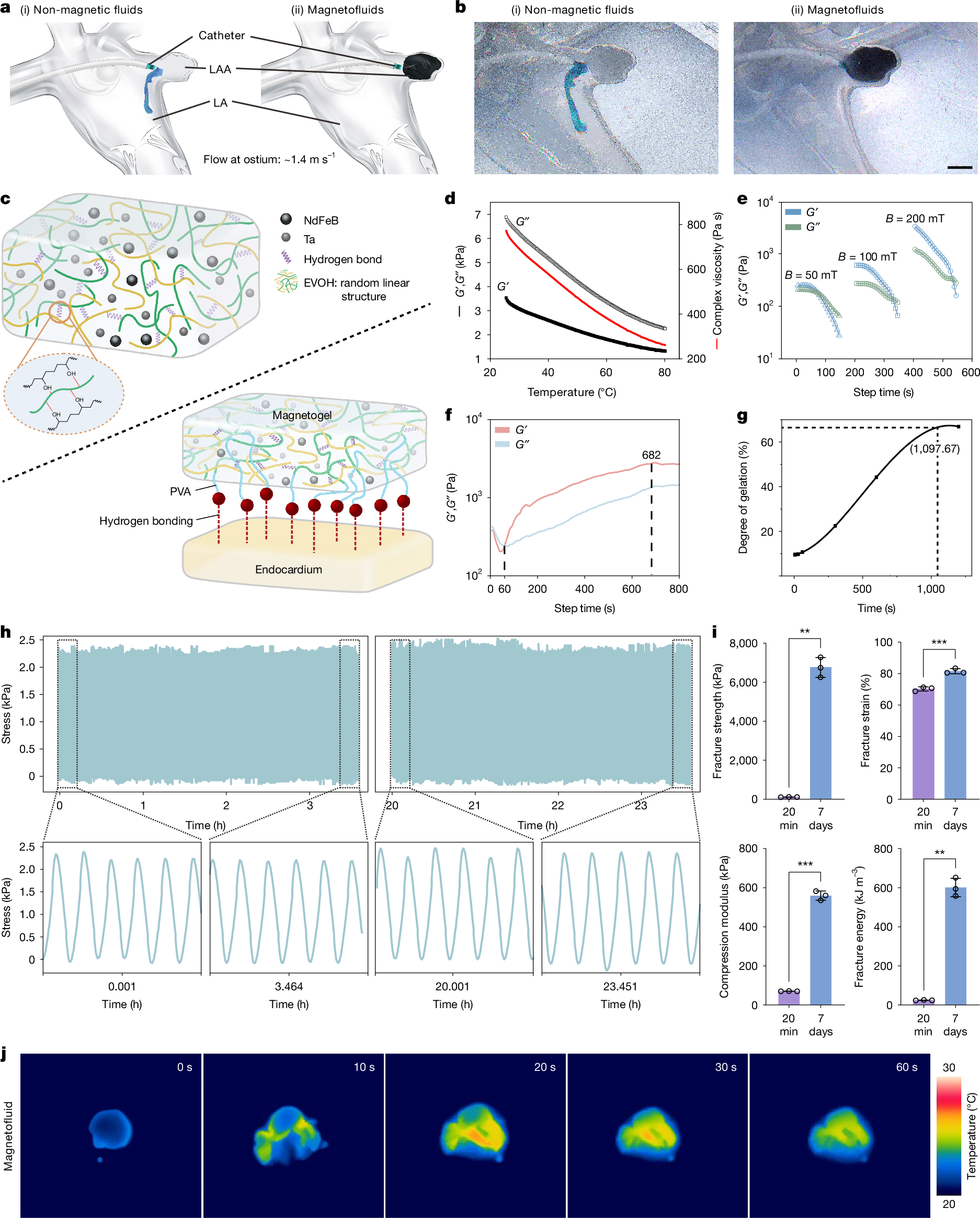
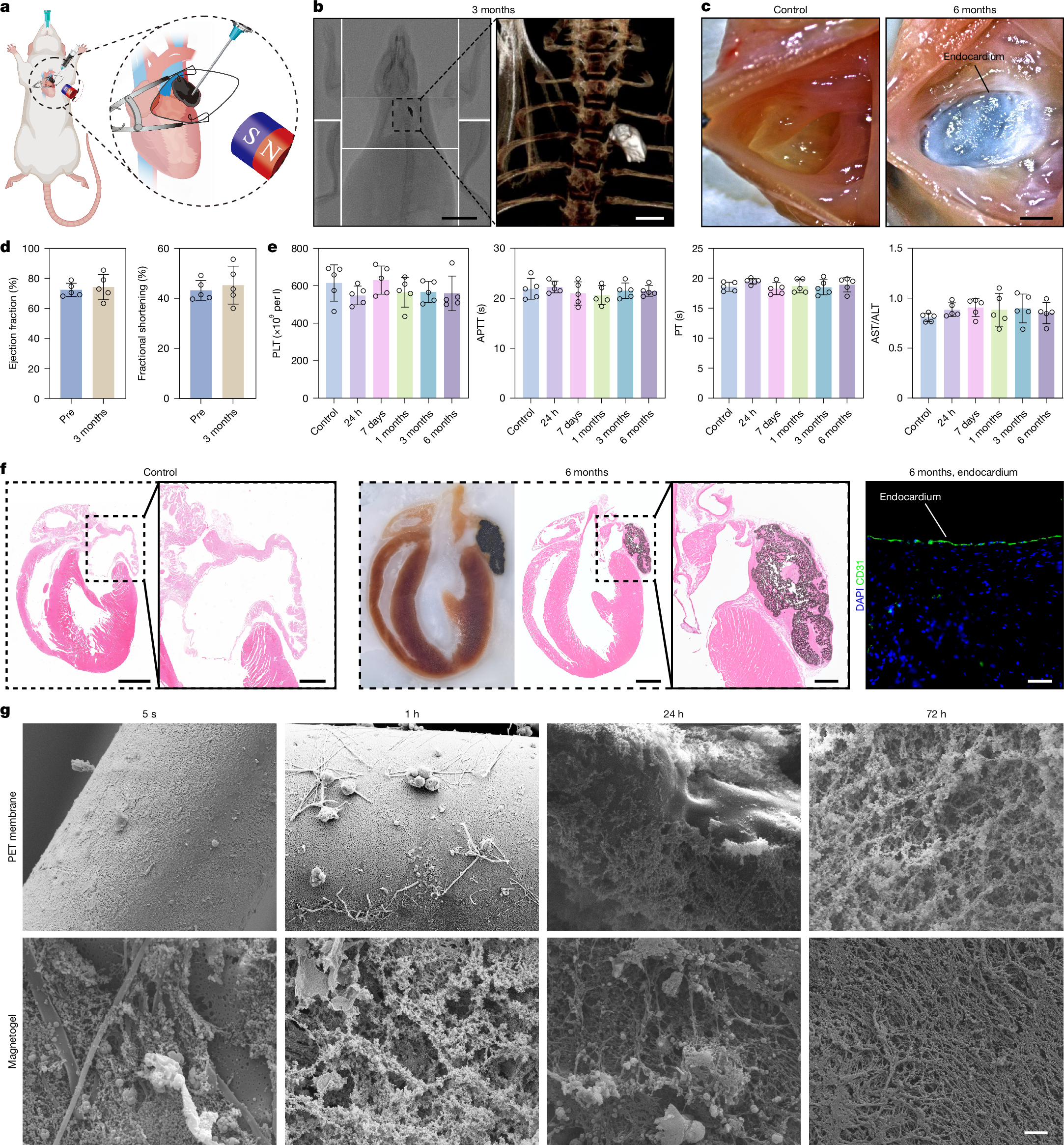
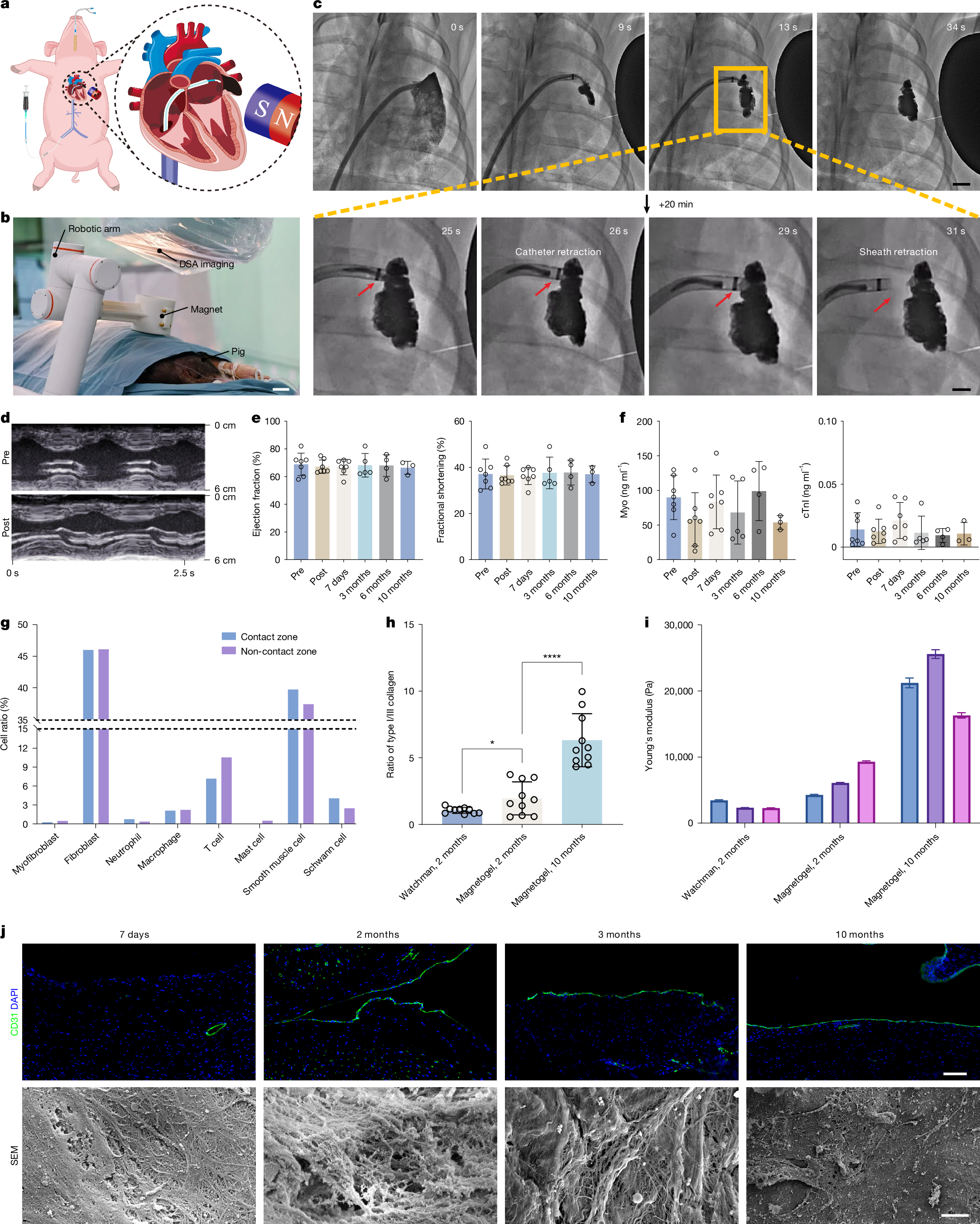
Peri-device leak and device-related thrombus1 remain key challenges of current left atrial appendage occlusion (LAAO)2 owing to the incompatibility between the solid occluder and the left atrial appendage (LAA). Here we propose a personalized and complete LAAO using magnetofluids that is suitable for all types of LAAs. Magnetofluids can be injected into LAAs from cardiac catheters. In the presence of a sufficient magnetic field, magnetofluids can resist high-speed blood flow. Magnetofluids can precipitate into magnetogels in contact with water in the blood within only a few minutes. We further confirmed the long-term resilience and biocompatibility of the magnetogel over 10 months in a pig model in vivo. Neither device-related thrombus nor magnetogel leakage was observed in any pigs. The endocardium formed on the Watchman occluder was rough and incomplete, predisposing to thrombosis. Myocardial injuries were unavoidable due to the barbs of the Watchman occluder. The endocardium formed on our magnetogel was smooth, firm and thrombus-free. No crevice was observed between our magnetogel and the LAA, and no injury was caused to the myocardium. These findings may offer a promising clinical strategy for long-term thrombus-free LAAO.
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout
Data availability
scRNA-seq data have been deposited in a public repository (ArrayExpress: E-MTAB-15847). The Sus scrofa (pig) reference genome (Sscrofa11.1) can be found at NCBI (GCF_000003025.6). Other original data including the animal-based dataset and retrospective clinical dataset are available at GitHub (https://github.com/rinrpg/Origin-data-of-Long-term-thrombus-free-left-atrial-appendage-occlusion-via-magnetofluids). Authentication documents of the cell lines are available at GitHub (https://github.com/silver2008a5/Cell-line-authentication-documents.git). The animal illustrations in Figs. 3a and 4a and the cardiac illustrations in Fig. 4a and Supplementary Fig. 41a were adjusted using BioRender. The original illustrations can be downloaded from the following links: models of rat and its heart in Fig. 3a (https://BioRender.com/yl2wllu); pig model in Fig. 4a (https://BioRender.com/g716ki6); and cardiac models in Fig. 4a and Supplementary Fig. 41a (https://BioRender.com/3a9pht3). Source data are provided with this paper.
Code availability
The scRNA-seq analysis codes from this study have been deposited at GitHub (https://github.com/pkulhc/Single-Cell-RNA-Seq-Analysis).
References
Alkhouli, M. et al. Left atrial appendage occlusion: current advances and remaining challenges. JACC Adv. 1, 100136 (2022).
Holmes, D. R. et al. Left atrial appendage occlusion: opportunities and challenges. J. Am. Coll. Cardiol. 63, 291–298 (2014).
Smit, J. M. et al. Anatomical characteristics of the left atrium and left atrial appendage in relation to the risk of stroke in patients with versus without atrial fibrillation. Circ. Arrhythm. Electrophysiol. 14, e009777 (2021).
Lip, G. et al. Atrial fibrillation. Nat. Rev. Dis. Primers 2, 16016 (2016).
Marini, C. et al. Contribution of atrial fibrillation to incidence and outcome of ischemic stroke: results from a population-based study. Stroke 36, 1115–1119 (2005).
Sievert, H. et al. Percutaneous left atrial appendage transcatheter occlusion to prevent stroke in high-risk patients with atrial fibrillation: early clinical experience. Circulation 105, 1887–1889 (2002).
Whitlock, R. P. et al. Left atrial appendage occlusion during cardiac surgery to prevent stroke. N. Engl. J. Med. 384, 2081–2091 (2021).
Saw, J. Left atrial appendage occlusion. Nat. Rev. Cardiol. 21, 153–154 (2024).