Microbiota-mediated induction of beige adipocytes in response to dietary cues
TL;DR
Low-protein diets induce white adipose tissue browning via gut microbiota, mediated by bile acids activating FXR in adipocytes and ammonia driving FGF21 in hepatocytes. This reveals microbiota-dependent pathways linking diet to metabolic remodeling.
Key Takeaways
- •Low-protein diets robustly induce browning of white adipose tissue, similar to cold exposure or β-adrenergic stimulation, in a microbiota-dependent manner.
- •Microbiota-derived bile acids activate the farnesoid X receptor (FXR) in adipose progenitor cells, while commensal ammonia drives fibroblast growth factor 21 (FGF21) expression in hepatocytes, both essential for browning.
- •Germ-free mice show diminished browning responses, which can be rescued by colonization with specific bacterial consortia from low-protein diet-fed mice or humans with brown/beige fat activity.
- •The browning effect is adipose-depot-specific, reversible, and modulated by factors like age, sex, and dietary context, with essential amino acid restriction playing a key role.
- •These findings uncover orthogonal pathways connecting diet, microbial metabolism, and host signaling to promote metabolic health through adipose tissue remodeling.
Tags
Abstract
Interactions between diet and the gut microbiota are fundamental to metabolic health, shaping energy balance and disease susceptibility1,2,3,4,5. However, the underlying mechanisms by which dietary and microbial factors converge to regulate host physiology remain unclear. Here we show that protein availability profoundly modulates the functional landscape of the gut microbiota and promotes remodelling of white adipose tissue (WAT). Specifically, low-protein diets (LPDs) robustly induce signature genes of browning in WAT to a similar extent to that seen in response to classical stimuli, such as cold exposure or β-adrenergic receptor activation6,7,8. LPD-mediated browning was markedly diminished in germ-free mice, and this defect was rescued by colonization with defined bacterial consortia made up of strains that were isolated and down-selected from the faeces of either LPD-fed mice or healthy human volunteers with 18F-fluorodeoxyglucose positron emission tomography (FDG-PET)-confirmed brown- or beige-fat activity9,10,11,12. Microbiota-induced browning was mediated both by bile acids driving the activation of the farnesoid X receptor (FXR) in adipose progenitor cells, and by nrfA-encoding commensal-derived ammonia driving the expression of fibroblast growth factor 21 (FGF21) in hepatocytes. The bile acid–FXR and ammonia–FGF21 axes both have non-redundant, essential roles in promoting WAT browning. These findings highlight a mechanistic link between diet, gut microbial metabolism and adipose tissue remodelling, uncovering microbiota-dependent pathways by which the host responds to dietary cues.
Main
Diet is a fundamental regulator of host physiology, influencing metabolism, immunity and susceptibility to a wide range of diseases1,2,3,4,5. The gut microbiota has emerged as a key mediator of dietary effects, contributing to interindividual variation in the nature, direction and magnitude of responses to nutritional and molecular inputs13,14,15. Among the many physiological processes that are influenced by diet–microbiota interactions, regulation of adipose tissue remodelling has emerged as an intriguing yet poorly characterized area. Unlike brown adipose tissue (BAT), which is enriched in mitochondria and expresses thermogenic proteins, such as uncoupling protein 1 (UCP1), WAT stores excess energy. However, notably, WAT retains considerable plasticity and can acquire brown-fat-like properties under specific conditions, such as cold exposure and catecholamine stimulation. This process, termed browning or beiging, gives rise to cells characterized by enhanced mitochondrial biogenesis and expression of thermogenic and lipid-oxidation-related genes6,16,17,18. Several dietary interventions—including caloric restriction, ketogenic diets and deprivation of methionine, leucine or cysteine—have been reported to activate BAT and induce WAT browning19,20,21,22,23,24. The gut microbiota is thought to have a role in mediating these responses, potentially through the production of bioactive metabolites and immunomodulatory effects25,26,27,28,29,30. However, which members of the microbiota are involved in particular dietary contexts, and how diet–microbiota interactions are translated into signals that drive beige-fat biogenesis, remain poorly defined. In this study, we identify two orthogonal pathways that link low protein intake, specific microbial metabolic activity and host signalling networks to promote WAT browning.
LPDs promote WAT browning
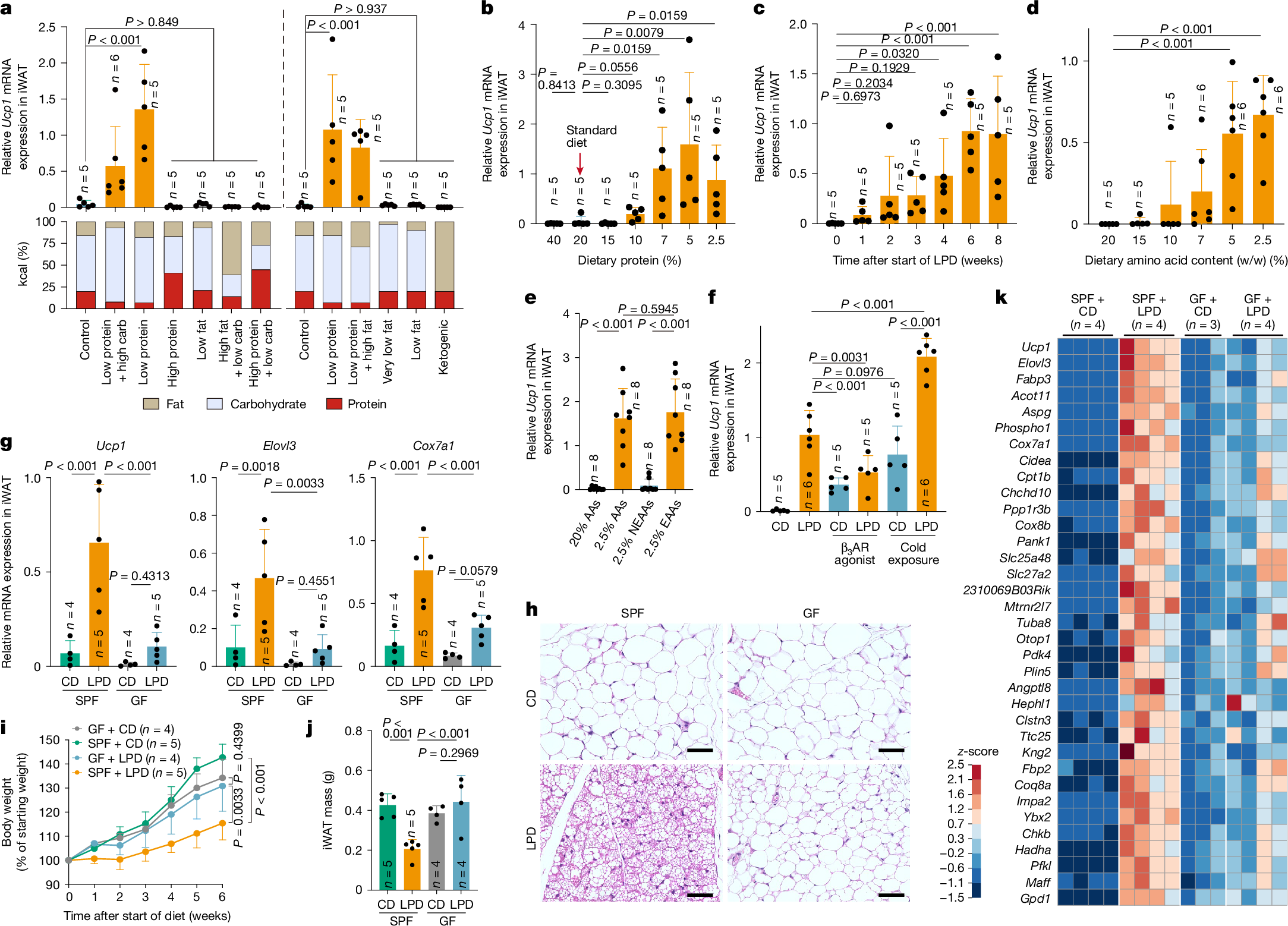
We first examined the effects of dietary modifications on WAT browning by formulating diets with varying proportions of protein, carbohydrate and fat. Although changes in fat content affected the total calorie count, all other diets were designed to be isocaloric. These diets were fed to specific-pathogen-free (SPF) C57BL/6 (B6) mice housed at room temperature (23 °C). Regardless of the lipid and carbohydrate content, feeding mice diets with low levels of protein significantly increased the expression of Ucp1 and Cox7a1 mRNA in the inguinal WAT (iWAT) (Ucp1 expression is shown in Fig. 1a, with Cox7a1 in Extended Data Fig. 1a. Throughout the manuscript, Ucp1 data are shown in the main figures and additional beige markers are provided mainly in the Extended Data). mRNA expression was assessed using three distinct methods31 with concordant results (Supplementary Fig. 1a). Feeding mice an LPD resulted in decreased iWAT mass (Supplementary Fig. 1b) and increased histological features of browning, including cells with multilocular lipid droplets, particularly in the central region adjacent to the inguinal lymph node (Extended Data Fig. 1b). B6 mice that were fed diets with a graded protein content showed a robust induction of Ucp1, Elovl3 and Cox7a1 in iWAT when dietary protein was reduced to 7% or less (a level approximately 60% lower than that of the control diet) (Fig. 1b and Extended Data Fig. 1c–e). Time-course analysis showed that these genes were induced within 2 weeks on a 7% protein diet and plateaued by weeks 6–8 (Fig. 1c, Extended Data Fig. 1f and Supplementary Fig. 1c). RNA sequencing (RNA-seq) analysis revealed that a broad range of beige-fat signature genes were upregulated in iWAT, including those involved in mitochondrial function (such as Cox7a1 and Cox8b), thermogenesis (such as Elovl3, Dio2 and Ucp1) and lipid metabolism (such as Cidea, Dio2, Cpt1b and Acott11); this upregulation began within 1 week of LPD feeding and became progressively more pronounced over the next 6 weeks (Extended Data Fig. 1g,h). The iWAT transcriptional profile induced by LPD feeding closely resembled that induced by a β3-adrenergic receptor agonist (Extended Data Fig. 1h). A reduction in dietary protein content also led to an increase in Ucp1, Elovl3 and Cox7a1 expression in interscapular BAT, although at lower magnitudes than that observed in iWAT (Extended Data Fig. 1i).
a, SPF B6 male mice were fed diets with varying proportions of protein, carbohydrate (carb) and fat for 6 weeks. Ucp1 mRNA expression in iWAT, normalized to Ppib, is shown together with each diet’s macronutrient composition. The left and right panels represent two independent experiments. b–e, Relative Ucp1 mRNA expression in iWAT in mice that were fed isocaloric diets containing the indicated protein concentrations for 6 weeks (b), a 7% LPD for the indicated durations (c), diets containing defined amino acids with varying total amino acid contents for 6 weeks (d) or diets containing 20% or 2.5% total amino acids (AAs), or with either EAAs or NEAAs reduced to 2.5%, for 6 weeks (e). f, SPF mice were fed a control diet (CD) or an LPD for 6 weeks and, during the final week, either received daily intraperitoneal injections of a β3-adrenergic receptor (β3AR) agonist (20 µg per mouse per day) or were exposed to cold (6 °C). g–k, SPF and GF mice were fed a CD or an LPD for 6 weeks. Ucp1, Elovl3 and Cox7a1 expression in iWAT (g), representative haematoxylin and eosin (H&E)-stained sections (h), body-weight change (i) and iWAT mass (j) are shown. Scale bars, 50 μm (h). k, Heat map showing genes enriched in iWAT of SPF + LPD mice compared with SPF + CD, GF + CD and GF + LPD mice (reads per kilobase per million reads (RPKM) ≥ 200, fold change ≥ 4, adjusted P < 0.05 with Benjamini–Hochberg correction). Each circle in the bar graphs represents an individual mouse. Data are mean ± s.d. Statistical tests: one-way ANOVA with Benjamini–Hochberg correction (a,c–g,j), two-tailed Mann–Whitney test for each comparison (b) and two-way ANOVA with Benjamini–Hochberg correction (i).
Feeding B6 mice a 7% protein diet for 6 weeks reduced body weight and whole-body fat volume, and enhanced glucose tolerance (Extended Data Fig. 2a–e). Mice showed no overt changes in appearance or behaviour (Supplementary Video 1), and did not exhibit increased faecal energy content or impaired lipid absorption (Extended Data Fig. 2f,g). In addition, whole-body lean volume and gastrocnemius muscle mass were mostly preserved (Extended Data Fig. 2h–j and Supplementary Video 2). These results indicate that the 6-week, 7% protein diet was generally well-tolerated and that the observed upregulation of browning markers and metabolic effects are unlikely to be driven mainly by pathological malabsorption or severe muscle wasting. Accordingly, this LPD protocol was used for all subsequent experiments, unless otherwise noted.
LPD feeding robustly induced signature genes of beige fat, even under thermoneutral conditions (30 °C) (Extended Data Fig. 2k). The LPD-induced formation of beige adipocytes in iWAT was observed across several mouse strains, including BALB/c and ICR, and was therefore not specific to B6 mice (Supplementary Fig. 2a). However, female or aged B6 mice showed attenuated browning, compared with male or young mice, respectively (Extended Data Fig. 2l,m). In addition, perigonadal WAT did not exhibit an induction of beige marker genes (Supplementary Fig. 2b). After reversion to a regular diet, beige marker expression and beige-adipocyte-like morphology declined (Extended Data Fig. 2n), indicating that LPD-induced browning in iWAT is both inducible and reversible. Increased expression of Ucp1 and metabolic effects were also observed when mice were switched from a high-fat diet (HFD) either to an LPD (Extended Data Fig. 3a–e) or to a high-fat and low-protein diet (HFD to HF/LPD; Extended Data Fig. 3f–j), although the magnitude of this induction was attenuated with prolonged or greater fat exposure. Collectively, these results indicate that LPD-induced browning is robust but adipose-depot-specific, and modulated by age, sex and dietary context.
Because protein type and digestibility might influence outcomes, we next examined diets containing defined amino acids instead of natural proteins. Decreasing the dietary amino acid content led to a marked increase in beige marker expression in iWAT, with maximal induction, comparable to that observed with a 7% LPD, achieved at 5% or less amino acids (Fig. 1d and Extended Data Fig. 3k). We then formulated diets with decreased levels of essential amino acids (EAAs) or non-essential amino acids (NEAAs). Reducing EAAs recapitulated the induction of beige marker genes that was observed with total amino acid limitation, whereas restricting NEAAs had no discernible effect (Fig. 1e and Extended Data Fig. 3l). We next examined the effect of reducing each individual EAA in the diet. Reduction of isoleucine, leucine, phenylalanine, tryptophan, lysine, methionine or threonine resulted in modest and variable inductions of browning (Extended Data Fig. 3m). These results suggest that although restriction of individual EAAs has limited effects, concurrent restriction of several EAAs engages complementary pathways to drive a more robust browning response.
LPD-induced browning requires the microbiota
We next investigated the mechanistic underpinnings of LPD-mediated iWAT browning. Feeding mice an LPD robustly induced the expression of Ucp1, Elovl3 and Cox7a1, to levels comparable with those induced by cold exposure (6 °C for 7 days) or treatment with a β3-adrenergic receptor agonist (Fig. 1f and Supplementary Fig. 3a). Notably, combining an LPD with cold exposure resulted in an additive increase in the expression of beige marker genes, suggesting that distinct—although possibly overlapping—mechanisms of browning are involved. Although immune cells have been implicated in cold-induced browning32,33,34,35,36,37,38, LPD-induced browning was preserved across several immune-deficient mouse models, including those lacking αβ and γδ T cells, B cells, innate lymphocytes, type 1, 2 or 17 immune molecules, myeloid cells or lymphoid tissues (Extended Data Fig. 4a). We therefore focused on immune-independent mechanisms.
We next assessed the role of the gut microbiota in LPD-induced browning using germ-free (GF) mice. Compared with SPF mice, GF mice exhibited significantly reduced expression of Ucp1,Elovl3, and Cox7a1 and fewer histological indicators of iWAT browning after being fed an LPD (Fig. 1g,h). In addition, although the LPD suppressed weight gain and WAT mass in SPF mice, no such effect was seen in GF mice (Fig. 1i,j). Low-level induction of Ucp1 was occasionally observed in LPD-fed GF mice, suggesting that a microbiota-independent pathway exists, although this response was not robust (Supplementary Fig. 3b). RNA-seq of iWAT from LPD-fed GF mice revealed a reduction in the expression of several beige signature genes, compared with LPD-fed SPF mice, including Ucp1, Cidea, Elovl3, Cox7a1, Cox8b, Clstn3 and Acot11 (refs. 39,40,41) (Fig. 1k). Gene Ontology enrichment analysis highlighted the reduced expression of genes involved in mitochondrial biogenesis and lipid metabolism in LPD-fed GF versus SPF mice (Supplementary Fig. 3c). Similar trends were observed in mice that were fed a low-EAA diet (Supplementary Fig. 3d). Treating adult SPF mice with an antibiotic cocktail also led to a significant reduction in the expression of Ucp1, Elovl3 and Cox7a1 in iWAT (Supplementary Fig. 3e). These findings indicate that the microbiota is a key driver of LPD-mediated beige-adipocyte induction.
LPD–microbiota–FXR pathway in browning
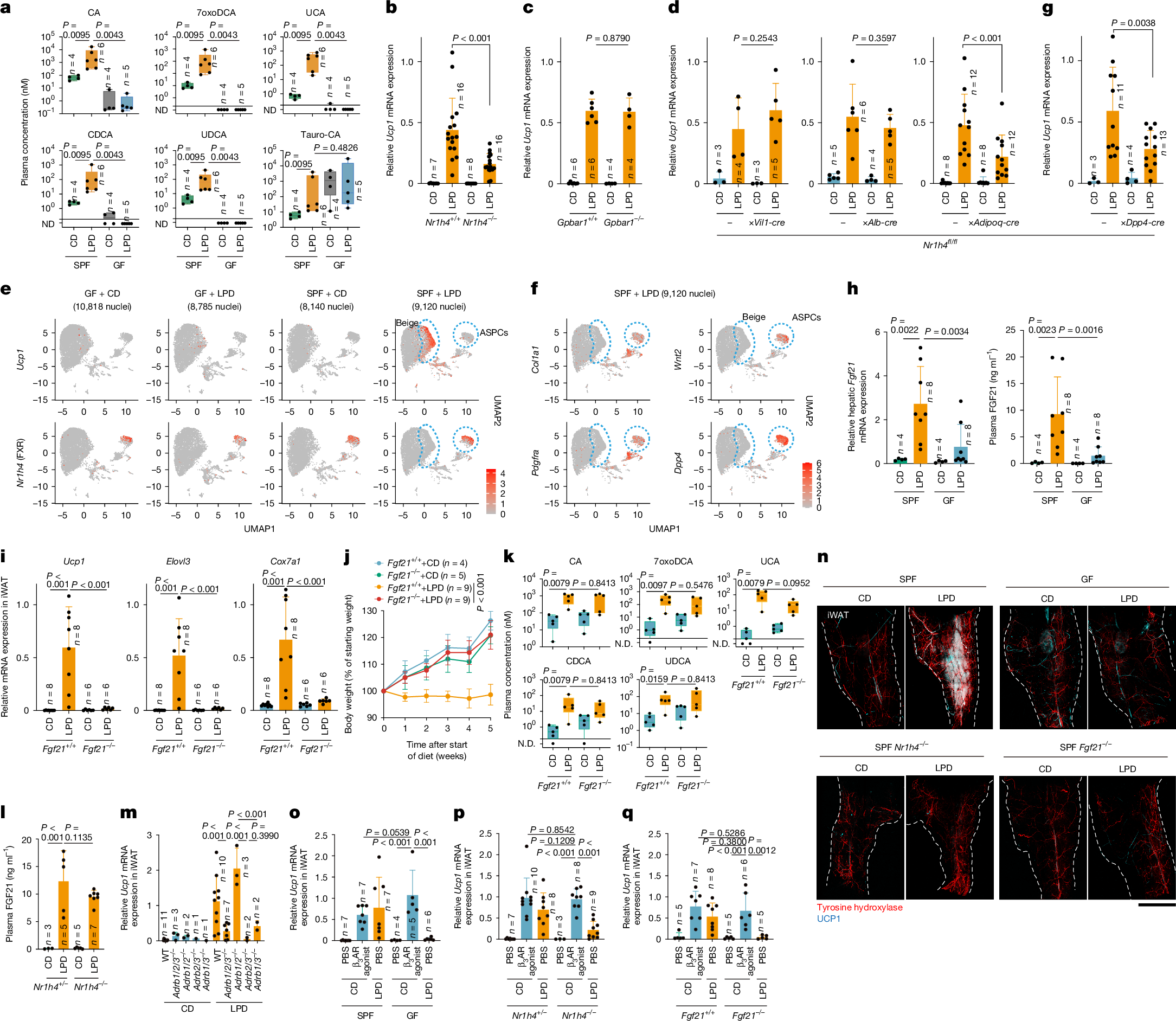
We next aimed to identify microbiota-derived molecules that might contribute to iWAT browning. Non-targeted liquid chromatography–mass spectrometry (LC–MS) analysis of ileal and plasma samples from SPF and GF mice that were fed a control diet or an LPD revealed that an LPD increased the plasma levels of unconjugated bile acids, including cholic acid (CA) and muricholic acid (MCA), in SPF mice (Extended Data Fig. 4a). Subsequent targeted LC–MS analysis confirmed the increased plasma levels of CA, αMCA, βMCA and chenodeoxycholic acid (CDCA), as well as 7α- and 7β-dehydroxylation products42, including 7-oxo-deoxycholic acid (7oxoDCA), ursocholic acid (UCA) and ursodeoxycholic acid (UDCA), in LPD-fed SPF mice compared with GF mice and control-diet-fed SPF mice (Fig. 2a and Extended Data Fig. 4b), although no such trends were seen in the intestinal contents (Supplementary Fig. 4a). These bile acids exhibited agonistic activity in an in vitro FXR reporter assay, indicating activation of this bile acid receptor (Supplementary Fig. 4b). Therefore, we next investigated whether FXR signalling was involved in browning. Mice deficient in FXR (encoded by Nr1h4) exhibited a significantly reduced induction of Ucp1 and Elovl3, fewer morphologically beige cells in iWAT and increased weight gain during LPD feeding, compared with wild-type controls (Fig. 2b, Extended Data Fig. 4c–e and Supplementary Fig. 4c). By contrast, deletion of another prominent bile acid receptor, TGR5 (Gpbar1−/−), did not affect LPD-induced browning (Fig. 2c, Extended Data Fig. 4d,e and Supplementary Fig. 4c). These findings indicate that microbiota-derived bile acids promote beige-adipocyte induction during LPD feeding at least in part through an FXR-dependent, TGR5-independent pathway.
a, Plasma bile acids in GF or SPF mice fed a CD or an LPD were quantified by LC–MS/MS. ND, not detected. b–d,g, SPF mice of the indicated genotypes were fed a CD or an LPD for 6 weeks, and iWAT Ucp1 expression, normalized to Ppib, was measured. e,f, Uniform manifold approximation and projection (UMAP) plots of iWAT snRNA-seq data for Ucp1 and Nr1h4 (e) and for Col1a1, Wnt2, Pdgfra and Dpp4 (f) from GF or SPF mice fed a CD or an LPD. Each dot represents a nucleus, and red indicates expression of the indicated gene, with colour intensity reflecting expression levels. Beige and ASPC clusters are outlined by blue dashed circles. h, Hepatic Fgf21 mRNA expression and plasma FGF21 levels in GF or SPF mice fed a CD or an LPD. i–k, Fgf21+/+ and Fgf21−/− littermates fed a CD or an LPD were analysed for iWAT gene expression (i), body-weight changes (j) and plasma bile acids (k). In k, n = 5 per group. l, Plasma FGF21 levels in Nr1h4−/− and Nr1h4+/− mice. m, iWAT Ucp1 mRNA expression in SPF mice of the indicated genotypes (Adrb1−/−Adrb2−/−Adrb3−/− (Adrb1/2/3−/−), Adrb1−/−Adrb2−/− (Adrb1/2−/−), Adrb2−/−Adrb3−/− (Adrb2/3−/−) or Adrb1−/−Adrb3−/− (Adrb1/3−/−)) after 6 weeks on a CD or an LPD. n, Representative whole-mount iWAT images stained for tyrosine hydroxylase and UCP1 with iDISCO processing; dashed lines indicate iWAT boundaries. Scale bar, 2 mm. o–q, Wild-type (WT) SPF and GF mice (o), or SPF mice of the indicated genotypes (p,q), were fed a CD or an LPD for 6 weeks and received a daily β3-adrenergic receptor agonist or phosphate-buffered saline (PBS) during the final week. Relative iWAT Ucp1 was measured. Circles represent individual mice; data are mean ± s.d. Box plots show median, interquartile range and range. Statistical analyses: one-way ANOVA with Benjamini–Hochberg correction (b–d,g–i,l,m,o–q), two-way ANOVA with Benjamini–Hochberg correction (j) and two-tailed Mann–Whitney test (a,k).
Given the broad expression of FXR in the intestine, liver and WAT43, we used tissue-specific Nr1h4-knockout mice to define the organ-specific contribution of FXR signalling to iWAT browning. Deleting FXR in intestinal epithelial cells (Nr1h4fl/fl;Vil1-cre) or hepatocytes (Nr1h4fl/fl;Alb-cre) did not affect LPD-induced Ucp1 expression. By contrast, adipocyte-specific deletion of FXR (Nr1h4fl/fl;Adipoq-cre) significantly reduced Ucp1 and Elovl3 expression and morphologically beige cells in iWAT (Fig.